When tau tangles, RNA errors can happen in Alzheimer’s disease
The tau protein is a key player in Alzheimer’s disease. It tangles and accumulates inside brain cells and this, in combination with increasing cell death, is a hallmark of the condition. Tau tangles seem to interfere with neuron’s cell functions in various ways.
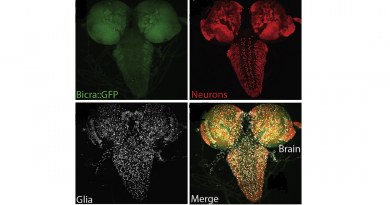
For instance, a discovery by the laboratory of Dr. Joshua Shulman and his colleagues revealed that triggering the formation of tau tangles resulted in the activation of fruit fly transposable elements. The findings strongly suggested that tau aggregates that disrupt the architecture of the genome can potentially mediate the activation of transposable elements and ultimately cause neurodegeneration.
The Shulman lab recently reported that tau tangles also seemed to interfere with other cellular process, leading to neurodegeneration.

“We looked into the possibility that tau tangles within neurons interfered with RNA splicing,” said Shulman, associate professor of neurology, neuroscience and molecular and human genetics at Baylor College of Medicine and investigator at the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital.
RNA splicing is involved in the maturation of RNA molecules necessary to produce working proteins. Alterations in RNA splicing are known to be involved in the development of certain neurodegenerative conditions, such as spinal muscular atrophy and amyotrophic lateral sclerosis. However, until now, their role in Alzheimer’s disease was not studied in great detail.
To carry out RNA splicing, cells use the spliceosomal complex, a multiprotein cellular machinery that coordinates the production of mature RNA molecules. RNA splicing is one of the important ways by which organs generate different cell types, each of which performs specialized functions and is especially critical to generate the cellular diversity and complexity in the human brain. Minor perturbations in the assembly and/or function of the spliceosome are predicted to render brain cells vulnerable to degeneration and premature death, particularly in aging individuals.
Recent studies using postmortem human brain tissue show that multiple components of the spliceosomal complex can bind to and co-aggregate with tau tangles. In the current study, the researchers began by overexpressing toxic levels of tau in fruit flies as a model to test whether spliceosome-tau interactions might cause neurodegeneration.
Tau tangles with the spliceosomal complex
“Using genetic interactions and functional assays, Yi-Chen Hsieh and Caiwei Guo, graduate students in my lab, identified several spliceosomal genes that mediated tau toxicity,” Shulman said.
Since many spliceosomal proteins were present in low levels in the neurons of flies overexpressing toxic tau, the researchers proposed that tau tangles either disrupted proper assembly of the spliceosome or sequestered key components in the cytoplasm, away from the site of action in the nucleus. This model was further supported by experiments in which flies expressing toxic tau showed evidence of global disruptions in RNA splicing that resulted in thousands of erroneous RNAs.

Analyzing specific splicing errors that occur with low frequency in large datasets is a time-consuming and challenging task. To conduct this analysis, Shulman and his colleagues collaborated with computational scientists in the laboratory of Dr. Zhandong Liu, associate professor of pediatrics-neurology at Baylor and also an investigator at the Jan and Dan Duncan Neurological Research Institute, to automate this process.

“Identifying and classifying specific splicing errors turned out to be fairly difficult. Current computational tools that analyze RNA sequencing datasets typically filter out any change that does not match the normal RNA splicing patterns. We had to specifically look into this ‘junk material’ to identify patterns of specific splicing errors,” Liu said.
For this study, Dr. Hari Krishna Yalamanchili, a postdoctoral fellow in the Liu lab, modified CrypSplice, a novel computational tool he had developed earlier, to perform in-depth analysis of splicing errors, which uncovered a link between tau tangles and an increase in a particular type of splicing errors.
This is the first study to demonstrate the role of tau tangles in disrupting the cellular localization and function of spliceosomal complex components that result in global splicing errors and cause progressive loss of neurons,” Shulman said.
“Our findings present an exciting new possibility of using RNA splicing as a potential molecular target for Alzheimer’s disease and other tau-mediated neurodegenerative conditions.”
Interested in reading more about this work? Find it in the journal Cell Reports.
Other contributors of this work include Measho Abreha, Rami Al-Ouran, Yarong Li, Eric B. Dammer, James J. Lah, Allan I. Levey, David A. Bennett, Philip L. De Jager, Nicholas T. Seyfried. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital, Emory University School of Medicine, Rush Alzheimer’s Disease Center, Columbia University Medical Center and Broad Institute.
This work was supported by grants from the National Institutes of Health (R01AG036836, R01AG050631, R01AG057339, U01AG046161, P30AG10161, R01AG15819, R01AG17917, U01AG46152, R01GM120033, P30CA125123 & U54HD083092), the Huffington Foundation, the Burroughs Wellcome Fund, the Cancer Prevention Research Institute of Texas, Houston Endowment and Belfer Neurodegenerative Disease Consortium.