Team work and balance can keep a gut healthy
A significant body of work currently is indicating that the microbiota helps shape the immune system and allows it to do its job. Disease-causing microbes, such as Salmonella, evoke a strong inflammatory immune response that is directed at eliminating the microbe. But an inflammatory immune response, especially in the intestine, can be damaging to the healthy tissue. How can then the body balance the immune responses and protect the gut from unwanted inflammation?
In her lab at Baylor College of Medicine, Dr. Gretchen Diehl, assistant professor of molecular virology and microbiology and a member of the Dan L Duncan Comprehensive Cancer Center, and her colleagues are investigating the role the microbiota plays in modulating the immune response in a way that reduces the damage inflammation can do to the gut.
Previous work has shown that the microbiota is required for the immune system to fight Salmonella, for instance. Using antibiotic treatment to remove the microbiota in laboratory animals results in the animals not producing an effective immune response against the microbe.

“In addition, we have learned that an effective immune response also depends on having immune cells called antigen-presenting cells. These cells ‘tell’ other immune cells, called T cells, to mount an inflammatory response to fight the microbes,” Diehl said.
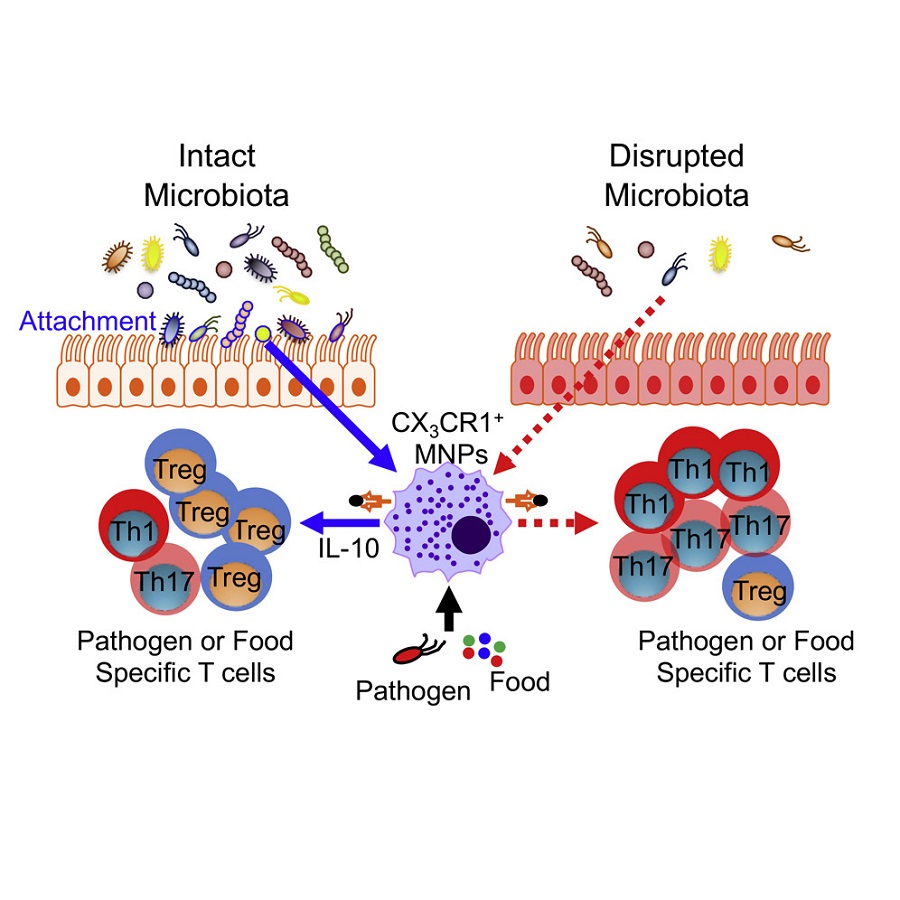
The microbiota also helps ‘tune down’ the inflammatory response by instructing the antigen-presenting cells to become anti-inflammatory. They instruct them to secrete the anti-inflammatory molecule called cytokine IL-10, which dampens inflammatory T cell responses and promotes regulatory T cell responses that keep the balance.
“This results in a balanced response that still can fight back Salmonella, but that is regulated to prevent damage to the healthy intestinal tissue,” Diehl said. “We wanted to know how the microbiota could induce these kind of responses.”
The microbiota-epithelium connection
“We found that when we gave the laboratory animals antibiotics, the antigen-presenting cells did not make IL-10. When we put back bacteria in the animals’ guts, only the bacteria that could attach to the intestinal epithelium were able to trigger IL-10 production by antigen-presenting cells and reduce the inflammatory response,” Diehl said. “It’s somewhat counterintuitive because microbes that can attach to the intestinal epithelium can potentially cause disease. But in this case we found that the attachment of bacteria to the epithelium was not causing damage; on the contrary, it was necessary to promote a balanced regulation of the T cell responses that would help protect the gut.”
The researchers indicate that their study is barely scratching the surface; there likely are other mechanisms by which microbes can promote a well-balanced intestinal environment.
Next, Diehl and her colleagues plan on investigating the signaling pathways that are activated when the microbes attach to the epithelial cells and result in triggering a balanced immune response.
“Another take home message is that a healthy microbiota is necessary to allow the immune cells to balance the responses that protect us from those that may harm us as they attempt to eliminate pathogens,” Diehl said. “Eating a diet abundant in whole grains, fruits and vegetables promotes the growth of good bacteria.”
The study suggests that manipulating the microbiota with the goal of modulating intestinal immune responses has potential therapeutic benefits for conditions such as irritable bowel disease.
Learn all the details of his study in the journal Immunity.
Other contributors to this work include Myunghoo Kim, Carolina Galan, Andrea A. Hill, Wan-Jung Wu, Hannah Felhner-Peach, Hyo Won Song, Deborah Schady, Matthew L. Bettini, Kenneth W. Simpson, Randy S. Longman and Dan R. Littman. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, New York University School of Medicine, University of Massachusetts Medical School, Texas Children’s Hospital, Cornell University, Weill Cornell Medicine and the Howard Hughes Medical Institute.
This study was supported by funding from the Curtis Hankamer Basic Research Fund at Baylor College of Medicine, the Texas Medical Center Digestive Disease Center Pilot Fund P30 DK056338 and National Institutes of Health grants AI123945, AI125264 and DK114252. Additional support was provided by the National Research Service Award T32AI053831_Corry, AAI Careers in Immunology Fellowship, the Howard Hughes Medical Institute, the Colton Center for Autoimmunity, the Texas Medical Center Digestive Disease Center Cellular and Molecular Morphology Core with funding from the NIH (P30DK056338) and the Cytometry and Cell Sorting Core at Baylor College of Medicine with funding from the NIH (P30 AI036211, P30 CA125123, and S10 RR024574).