Image of the Month: Molecular Hotspots
A handful of soil holds billions of microbes that interact with each other in many ways. Sometimes, microbes engage on chemical wars using antibiotics, which in turn prompt the production of defenses that counter the antibiotics. Not surprisingly, microbes then can develop defenses that neutralize the chemicals that provide resistance to antibiotics. And the war continues.
For instance, many bacteria produce β-lactamase, an enzyme that provides multi-resistance to β-lactam antibiotics such as penicillins and cephalosporins, among others. The soil bacterium Streptomyces produces β-Lactamase inhibitory proteins (BLIPs), which neutralize class A β-lactamases.
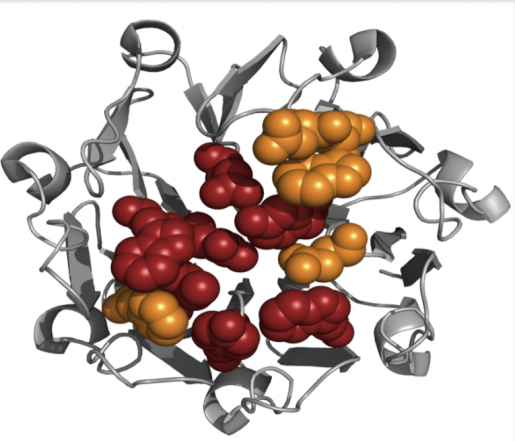
At Baylor College of Medicine, the lab of Dr. Timothy Palzkill studies the interactions between BLIPs and β-lactamases, looking for clues to understand what affects the attraction between these proteins. When BLIPs and β -lactamases bind to each other, their surfaces interact, but there are specific hotspots – specific amino acids or protein building blocks – that influence more than others the affinity of the interaction. These hotspots, when mutated to the amino acid alanine, can result in a greater than 10-fold decrease in binding affinity.
Improved insight into the factors influencing BLIP-β -lactamase binding not only provides a better understanding of protein-protein interactions, but also can have clinical implications.
Find all the information about this work in the journal Biochemistry.

Dr. Timothy G. Palzkill is chair and professor of pharmacology and chemical biology.