State of the Lester and Sue Smith Breast Center and breast cancer research
The Lester and Sue Smith Breast Center is a multidisciplinary center focused on breast cancer research and patient care. It is home to 19 tenure-track faculty members, including 13 scientists and 6 clinicians, as well as 42 postdoctoral associates and graduate students.

Dr. Xiang “Shawn” Zhang, interim director of the center and professor of molecular and cellular biology, sat down with From the Labs to provide an update on the state of the center and breast cancer research. Zhang is the William T. Butler, M.D., Endowed Chair for Distinguished Faculty, a McNair Scholar and a member of the Dan L Duncan Comprehensive Cancer Center at Baylor College of Medicine.

Q: What major questions are being explored in breast cancer research right now?
A: The majority of breast cancers are curable these days with surgical removal and adjuvant therapy, but there is still a good proportion of patients who develop a recurrence. That’s the major challenge for the entire breast cancer research field.
There are two questions at the heart of this. One is: Why does breast cancer recur and metastasize to other organs? The other question is: Why are some tumors resistant to therapy? These two questions are linked because often you have both metastasis and therapeutically resistant tumors. When tumors metastasize and recur, that means the cancer cell survived the original therapy.
All of our faculty members in the Breast Center are looking for new treatments. There are two ways to do that. One is to understand the mechanism of cancer and use existing drugs for treatment, either in new combinations or repurposing drugs not previously used in breast cancer. Those represent a faster path because the drugs are already tested. The other way is to create new drugs. That is a longer path, but the degree of innovation is higher.
Q: What are some major discoveries in the Breast Center that you are excited about?
A: The research in the center covers a wide spectrum. We approach the same questions from different angles. We have faculty using a proteomic approach – they look at proteins, which most directly show function and are the active target of drugs. The center is at the cutting edge of proteogenomic technology. We have profiled a lot of tumors before and after treatment, looking for new therapeutic targets. There are major findings along that front.
We also have faculty members using an integrated omics approach – they use data generated in the field in the past decade, including proteogenomic and transcriptomic data, to try to determine mechanisms of underlying treatment resistance.
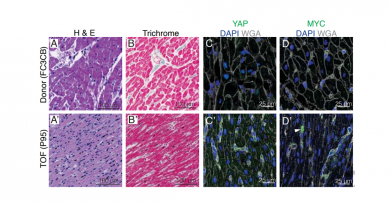
A more recent discovery involves proteins related to DNA repair and replication, the basic machinery of normal cells. When those proteins are mutated, they confer properties to tumors that may make them more vulnerable to certain therapies. 
In my own lab, we are trying to understand how tumors escape the immune system. We have zoomed into subpopulations of immune cells and are trying to understand the function of those immune cells in patients’ tumors that allow those tumors to progress and metastasize.
We also have research focusing on developing new animal models that can better mimic human disease. It’s always a challenge to completely replicate human disease properties. Those are incredibly important because without accurate models, you cannot do faithful preclinical studies. We have a wide spectrum of patient derived xenografts, tumors directly taken from patients, which more faithfully maintain patient characteristics. We also have new animal models like rat models. They have surprisingly more power replicating certain subtypes of breast cancer. To me, that’s a big advance in the field.
Q: What will this research mean for the future of patient care in the clinic?
A: Different research projects are at different stages. We are testing drugs that are already in the clinic; the question is how to better use them in combinations with other drugs or to identify the right group of patients for these drugs. We will see a near-term impact – better drugs, more precision and new combinations, in a few years’ time.
Some of this research is already in the clinic. A group in the center found that if you combine two drugs that had been individually used before, you can achieve a much better efficacy and eliminate the need for chemotherapy that had heavy side effects.
There also are research projects in the center that will have a big long-term impact. We are working to identify new drug targets. Those will need to undergo drug discovery and clinical trials.
Q: What is the next big thing in breast cancer research?
A: Personally, I am a fan of the immune system. Cancer has been proven to be very versatile and changeable, making it very difficult to cure. Previous types of therapies have suffered from that. When you have a targeted therapy, the tumor evolves to escape it and you see resistance after some time. The only thing that I think can match this evolvability is our immune system. We have seen numerous examples from other cancer types and preclinical studies. When the immune system works correctly, it can eliminate the tumor completely and achieve a durable response.
The current data in breast cancer clinical trials have not been able to convince everyone that immunotherapy is the answer for all breast cancer, but I think going forward, there will be better and better immunotherapies that will increase the success in breast cancer treatment.
By Molly Chiu
Interested in learning more about research projects at the Breast Center? Here are some of their recent publications:
- Dissemination of bone metastasis linked with bone remodeling
- Research reveals markers of chemotherapy resistance and outcome in triple negative breast cancer
- How breast cancer cells can become resistant to therapy
- A potentially more effective treatment for HER2 mutant metastatic breast cancer
- Combating triple-negative breast cancer on two fronts delivers promising results