A potentially more effective treatment for HER2 mutant metastatic breast cancer
Breast cancers with HER2 mutations respond to the drug neratinib, but the responses are variable and often not durable. Looking to better understand the underlying cause of this variation, a team led by researchers at Baylor College of Medicine investigated whether different HER2 mutations drove different responses to therapy.
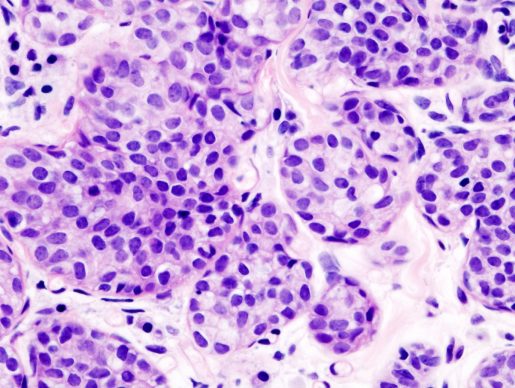
“In this study we focused on two types of breast cancer, ductal breast cancer and lobular breast cancer, which is understudied when compared to ductal breast cancer,” said leading author Dr. Shyam M. Kavuri assistant professor at Baylor’s Lester and Sue Smith Breast Center and the Department of Medicine. “More than 20% of patients with these types of breast cancer develop a metastatic or invasive form of the disease, for which there is no current treatment.”

The researchers looked into studies that sequenced the genes of human lobular and ductal breast cancers, specifically looking at the mutations in the HER2 gene. They found that the HER2 L755S mutation was more common among patients with metastatic lobular breast cancer than in ductal breast cancer patients.
Furthermore, patients with the HER2 L755 alterations had significantly reduced overall survival when compared to those carrying the natural or non-mutated form of HER2, suggesting that recurrent L755 alterations are associated with the more aggressive form of lobular breast cancer.
“On the other hand, we did not detect any difference of overall survival in ductal breast cancer with HER2 mutations, suggesting an important difference between the lobular and the ductal form with critical clinical implications,” said Kavuri, a member of the Dan L Duncan Comprehensive Cancer Center at Baylor.

To take a closer look into how this HER2 mutation influenced cancer behavior, the researchers conducted laboratory experiments in which the HER2 L755S gene was introduced into cells. Then, they treated the modified cells with drugs approved to treat the human cancer. “We found that having the HER2 L755S mutation conferred the cells resistance to treatments, including neratinib – the cells continued to grow,” said Dr. Rashi Kalra, first author of the study and a postdoctoral research associate in Kavuri’s laboratory. “When human tumor cells carrying this mutation were grown in animal models, they developed metastasis more often than cells not carrying this mutation.”
The findings suggest that the HER2 L755S mutation plays a role in the aggressiveness of lobular breast cancer observed in the clinic.
The search for a better treatment
Kavuri and his colleagues took a step further searching for drugs that could better treat cancer carrying the HER2 L755S mutation.
Of all the drugs we tested, poziotinib was able to completely inhibit both tumor growth and metastasis in our experimental models,” Kavuri said.

“We are excited by our findings,” said co-author Dr. Bora Lim, associate professor of oncology-medicine at Baylor and Director of Translational Research at Baylor’s Dan L Duncan Comprehensive Cancer Center. “Poziotinib offers the possibility of a more effective treatment option for HER2 mutant metastatic breast cancer. We are actively working on the development of a phase II clinical trial to determine the value of this drug in the treatment of patients with this devastating condition.”
Find all the details of this study in Cancer Research, a journal of the American Association for Cancer Research.
Other contributors to this work include Ching Hui Chen, Junkai Wang, Ahmad Bin Salam, Lacey E. Dobrolecki, Alaina Lewis, Christina Sallas, Clayton C. Yates, Carolina Gutierrez, Balasubramanyam Karanam, Meenakshi Anurag and Matthew J. Ellis. The authors are affiliated with Baylor College of Medicine or Tuskegee University.
This study was funded by Susan G. Komen (grant CCR16380599) and Department of Defense (grants W81XWH-18-1-0040 and W81XWH-18-1-0084), Susan G. Komen Promise grant PG12220321, Cancer Prevention and Research Institute of Texas (CPRIT) Recruitment of Established Investigators award RR140033 and SPORE grant P50CA186784-06. Further support was provided by P30 Cancer Center Support Grant NCI-CA125123, CPRIT Core Facilities Support Grant RP170691, NIH grant 1R41CA257110-0 and DOD/PCRP PC190741.