How genomics profiling can help identify the best treatment for bladder cancer
When it comes to cancer, one-size-does-not-fit-all.
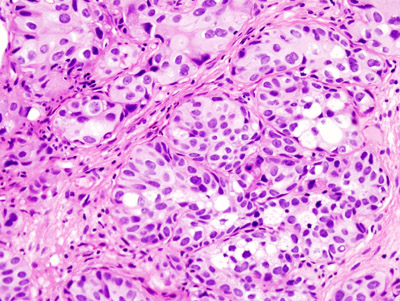
“One of the challenges that we have when taking care of patients with bladder cancer is that from one patient to the next, the prognosis, the stage and the response to different kinds of treatment differ,” said Dr. Seth Paul Lerner, who is the director of urologic oncology and of the Multidisciplinary Bladder Cancer Program as well as professor of urology and Beth and Dave Swalm Chair in Urologic Oncology at Baylor College of Medicine. “The diverse cancer characteristics pose a challenge when selecting the best treatment for each patient.”

At Baylor, Lerner and his colleagues have been studying the genomic underpinnings of these differences among patients with muscle invasive bladder cancer. As part of the Cancer Genome Atlas Research Network group, they reported in 2017 a comprehensive molecular characterization of 412 muscle-invasive bladder cancers that resulted in the identification of five expression-based cancer subtypes. The researchers also reported the different survival outcomes of each subtype. For instance, one of the subtypes named ‘neuronal’ has a very distinct expression profile and is associated with poor survival and less favorable outcomes.
“We were able to show that mutation signatures, molecular subtypes, load of new cancer-associated molecules and known clinical and pathological factors have a very clear influence on overall patient survival,” Lerner said. “But, how can we apply this knowledge into clinical practice?”
“In this study published in European Urology in 2019, we report the development of a computational tool – a single-patient classifier – that effectively enables physicians to assign a bladder cancer subtype to an individual patient’s cancer using that patient’s genomic data,” said first author Dr. Jaegil Kim, who was at the Broad Institute at the time he was working on this project and currently is a senior principal scientist in TESARO, an oncology-focused company.
The researchers also applied their single-patient classifier to the data produced by the IMvigor 210 clinical trials that treated nearly 400 patients with locally advanced or metastatic bladder cancer with atezolizumab, a type of immunotherapy drug, and reported objective response and overall survival. This enabled the researchers to connect response to treatment with a particular bladder cancer subtype.
One of the most exciting findings is that the aggressive neuronal expression subtype we had identified in 2017 as having poor survival and less favorable outcome responds very well to atezolizumab treatment and patients have a much better outcome,” Kim said.
“Of the 11 patients we identified as having a neuronal subtype, all of those evaluable for objective response responded to the treatment (two complete response, six partial response), or 72 percent overall. This translated to a very high survival probability which is unprecedented in advanced bladder cancer,” Lerner said. “Although this is a small group of patients, it is very exciting to see that our basic research can be directly translated to the clinical setting allowing us to determine which subtype of bladder cancer has a better chance to respond well to a specific treatment.”
Although further testing is needed in prospective clinical trials, the researchers anticipate a future in which a physician can apply the single-patient classifier to patients’ tumor genomic data to identify the subtype of bladder cancer, and then select the treatment the particular subtype is most likely to respond to.
“Bladder cancer causes an estimated 160,000 deaths worldwide per year and we are behind other cancer fields in terms of the clinical applications of its molecular data and biology,” Lerner said. “However, we can begin to see how we can use this information in the future to provide the best treatment for each patient.”
Other contributors to this work include David Kwiatkowski (Harvard Medical School), David J. McConkey (Johns Hopkins University), Joshua J. Meeks (Northwestern University), Samuel S. Freeman and Gad Getz (the Broad Institute of MIT and Harvard) and Joaquim Bellmunt (Dana-Farber Cancer Institute and Harvard University).
The authors declared no financial support for this work. For the authors’ financial disclosures follow this link.