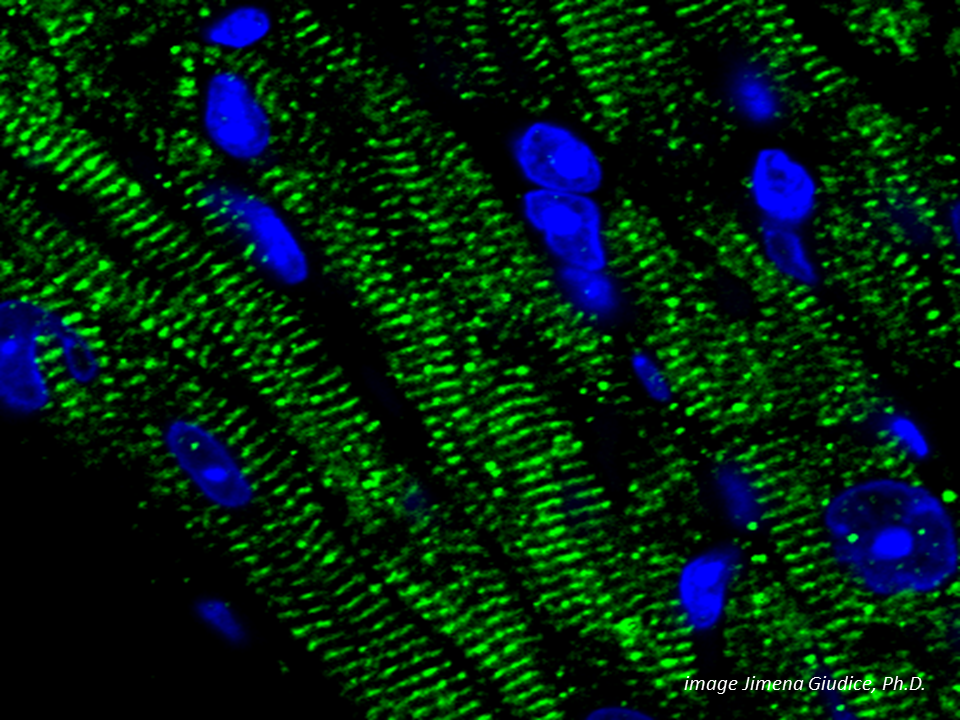
Eliminating p63 gene could convert scar tissue into heart muscle
After a heart attack, the parts of the heart muscle that die do not regenerate into new heart tissue; instead, they are replaced by scar tissue that does help the heart beat.

“Nearly 5 million Americans can be expected to develop advanced congestive heart failure, and, currently, heart transplant or mechanical circulatory support implantation are the only options for patients with end-stage heart failure,” said Dr. Todd Rosengart, chair and professor of the Michael E. DeBakey Department of Surgery at Baylor and senior author of the paper. “Our latest findings offer a promising new solution for treatment and improved cardiac function.”
Using rodent models, Rosengart and his colleagues have been looking for a means to genetically convert this scar tissue into muscle tissue at the cellular level, which could ultimately be a way to treat heart attack and heart failure patients.
The goal of the study was to increase the plasticity of the heart scar cells, meaning their ability to take on characteristics of heart muscle cells.
Through gene transfer studies in the lab, Rosengart and a team of researchers discovered that a gene called p63 appears to repress cell plasticity. When the researchers knocked out this gene, they found that cell reprogramming increased and some cells developed characteristics of heart muscle cells. Under proper conditions, other cells started beating.
This could potentially be a safe and effective strategy for inducing human cardiac cellular reprogramming as a potential therapeutic strategy for the treatment of heart failure,” said Rosengart, also holds the DeBakey-Bard Chair in Surgery at Baylor College of Medicine.
The next step in the research is to use this new strategy to improve heart function in animal models, which the researchers have already accomplished previously with other less potent reprogramming strategies.
Read all the details of this work in the in The Journal of Thoracic and Cardiovascular Surgery.
Others who took part in the research include Dr. Vivekkumar Patel, Dr. Vivek P. Singh, Jaya Pratap Pinnamaneni, Deepthi Sanagasetti, Jacqueline Olive, Megumi Mathison and Dr. Jianchang Yang with Baylor College of Medicine; Dr. Austin Cooney with Dell Medical School at The University of Texas at Austin; Dr. Elsa R. Flores with the Moffitt Cancer Center in Tampa, Fl.; Dr. Ronald G. Crystal with Weill Cornell Medical College.
The study was funded by the National Heart Lung and Blood Institute of the National Institutes of Health (1R01HL121294-01A1).