New insights into the fate of dying cells
An important part of an organism’s health is to effectively dispose of its dying cells. “Dying cells have to be eliminated fast before they break down and release their content into surrounding healthy tissue. Failing to clear up dying cells in time can induce direct tissue injury, inflammation, an autoimmune response, lead to chronic conditions or contribute to the spread of viruses growing inside them,” said Dr. Zheng Zhou, professor in the Verna and Marrs McLean Department of Biochemistry and Molecular Biology at Baylor College of Medicine.
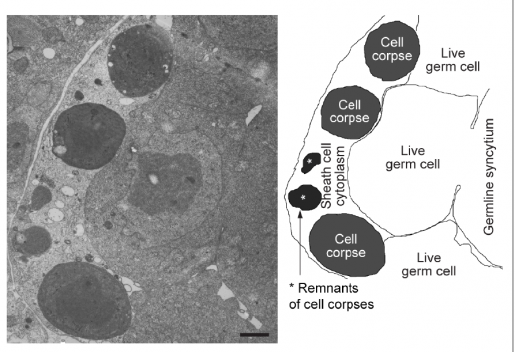
Housekeeping cells eliminate dying cells through a process called phagocytosis, which literally means ‘eating cells.’ Housekeepers known as phagocytes identify, engulf and ingest dying cells into vesicle-like structures called phagosomes, where they digest them to effectively eliminate them and recycle their components. The Zhou lab at Baylor has been conducting basic research on phagocytosis for many years, and in this study they provide key new insights into this essential process.
“We know that phagosomes cannot degrade dying cells on their own. They team up with other cellular structures called lysosomes, which fuse with the phagosomes and provide them with a battery of digestive enzymes, including those that break down proteins, DNA, lipids and sugars,” said Zhou, a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “Lysosomes keep their content under very acidic conditions, which is necessary for the enzymes to be active.”

Traditionally, lysosomes have been considered the primary source of digestive material for the phagosomes. Zhou and her colleagues wondered whether other intracellular structures also interacted with phagosomes to degrade dying cells.
Autophagy-phagocytosis crosstalk
There is another housekeeping process called autophagy that cells use to degrade and recycle their own damaged internal structures and proteins. This process involves autophagosomes, intracellular, double-membrane vesicle-shaped, enzyme-loaded structures that identify, ingest, digest and recycle damaged materials.
Until now, autophagy and phagocytosis were considered two parallel processes. Phagocytosis occurs when one cell eats another, while autophagy is a process inside a cell that eats cellular material. We have shown that rather than being independent processes, they cross talk, one contributes to the other,” said first author of the work, Omar Peña-Ramos, graduate student in the Zhou lab.

Using time-lapse fluorescence microscopy to measure dynamic changes in phagosomes, as well as electron microscopy, the researchers followed the degradation of dying cells in specific phagocytic cells in the embryos of the model organism Caenorhabditis elegans, a worm that is as long as a credit card is thick.
“We tracked a number of intracellular structures, autophagosomes included, looking if they also interacted with phagosomes to degrade dying cells,” Zhou said. “We found that autophagosomes actually fuse with phagosomes, contributing to the elimination of dying cells.”
“There are important benefits of using C. elegans as a model organism,” Peña-Ramos said. “The worm is transparent. It allows us to study biological processes, unlike in mammalian systems, by actually seeing the eating and degradation of dying cells in a living organism. Furthermore, the process takes place in about an hour. We can do many repeat experiments in a single day.”
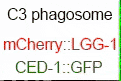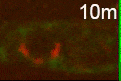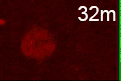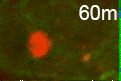
The researchers also found that C. elegans mutants that were defective in the production of autophagosomes displayed significant defects in the degradation of dying cells, demonstrating the importance of autophagosomes to this process.
The team also identified the molecular mediators of the autophagy-phagocytosis crosstalk. This involves the phagocytic receptor CED-1, which recognizes the surface ‘eat-me’ signal on a dying cell that promotes its ingestion. After ingestion is complete, under the order of CED-1, DYN-1, which is a large GTPase, promotes the recruitment of autophagosomes to phagosomes with the help of other molecules, which results in the digestion of the dying cell, the authors explained.
Further observations suggest that autophagosomes contribute to the degradation of dying cells, in addition to and in parallel of lysosomes,” Zhou said.
“Before, we thought that the autophagosome content was only for degradation for materials of the own cell, now we have shown that it also is used to clear other cells, specifically dying cells.”
Find the complete study in the journal eLife.
Other contributors to this work include Lucia Chiao, Xianghua Liu, Xiaomeng Yu, Tianyou Yao and Henry He, all at Baylor College of Medicine.
The National Institutes of Health grant NIH R01GM067848 supports this work.



