An organic insecticide is more damaging to non-target insects than synthetics
Large-scale insecticide application is a primary weapon in the control of insect pests in agriculture but we know that around the world, insect populations are decreasing in size by about 1% each year; this decrease is largely in insects that are not pests.

Spinosad is a popular organic insecticide commonly used to control insect pests, including thrips, leafminers, spider mites, mosquitoes, ants and fruit flies, in both commercial and domestic settings. Spinosad has been registered for use in pesticides by the U.S. Environmental Protection Agency (EPA) since 1997.
As a natural substance made by a soil bacterium, Spinosad is often thought to be less harmful to beneficial insects and is frequently used as an alternative to synthetic insecticides.
The current study, a collaboration between the Baylor College of Medicine, University of Melbourne, the Jan and Dan Duncan Neurological Research Institute (Duncan NRI) at Texas Children’s Hospital and the University of Texas, supports that organic insecticides are not as harmless as they are thought to be. The work also adds to a growing body of evidence indicating that insecticides are contributing to the global decline in population sizes of many beneficial insect species.
Low-level exposure to Spinosad damages fruit fly brains
The team worked with the laboratory fruit fly, Drosophila melanogaster, to investigate the effects of low-level exposure (0.2 parts per million) of Spinosad on the brain.
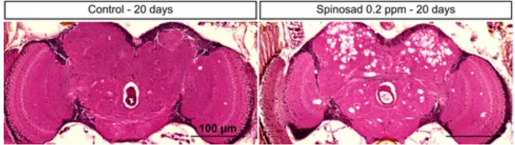
“Within a matter of 20 days, tiny doses of Spinosad can have alarming consequences on the brains of adult Drosophila. Observing sections of brain tissue under microscope revealed an average of 17% of the fly brains destroyed due to exposure,” said Dr. Felipe Martelli from Monash University, who completed this work as part of his Ph.D. at the University of Melbourne. “Neurons that serve vital functions die, leaving large vacuoles, fluid-filled sacs, in the brain. This leads to neurodegeneration, blindness and behavioral changes in adult fruit flies.”

Due to the Drosophila’s genetic and biochemical similarities to other insects, the researchers indicate that these impacts could be translated to other beneficial insects such as bees.
Martelli’s research was enhanced by the opportunity to do experiments in the lab of a global leader in neuroscience, Dr. Hugo Bellen, corresponding author and distinguished service professor of molecular and human genetics at Baylor and chair of neurogenetics at the Duncan NRI.

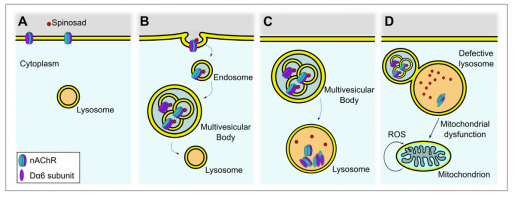
“The striking biological features that are associated with low levels of Spinosad resemble some slow progressive neurological diseases in which lysosomes are expanded, also called Lysosomal Storage Disease. These genetic diseases also have been associated with Parkinson’s Disease, and there are interesting similarities in the damaging mechanisms caused by mutations in these genes and Spinosad,” Bellen said.
Having an organic label doesn’t always mean safer
“There is often an assumption that organic equates to safer, but our study finds this isn’t the case,” said study co-author, Professor Philip Batterham, from the School of BioSciences and Bio21 Institute at the University of Melbourne.
“Spinosad is now registered for use in over 80 countries, and it poses a far greater risk to beneficial insects than previously thought. Concerningly, the low concentration levels used in this study is what would be commonly found in groundwater or in the air through incidental exposure.”
“Based on earlier work by our research group using similar techniques to this study, Spinosad was found to have a much greater negative consequences on fruit flies at far lower doses than imidacloprid, a synthetic insecticide that has been banned in Europe for its effects on non-target insects including honeybees,” Martelli said.

“While this study does not aim to pin the blame on Spinosad, it does show that having an organic label doesn’t always mean safer. All insecticides, no matter their source, need to be rigorously studied for any unintended ecological impacts,” Batterham said.
Read all the details of this study in the journal eLife.
Other contributors to this work include Natalia H Hernándes, Zhongyuan Zuo, Julia Wang, Ching-On Wong, Nicholas E Karagas, Ute Roessner, Thusita Rupasinghe, Charles Robin, Kartik Venkatachalam and Trent Perry. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, the University of Melbourne, McGovern Medical School at the University of Texas Health Sciences Center and the Jan and Dan Duncan Neurological Research Institute at Texas Children Hospital.
This work was supported by a Victorian Latin America Doctoral Scholarship, an Alfred Nicholas Fellowship, a UofM Faculty of Science Travelling Scholarship and the Robert Johanson and Anne Swann Fund. Further support was provided by the University of Melbourne, the Howard Hughes Medical Institute and an NIH (NIA) grant.