The bone microenvironment ‘empowers’ cancer cells to become metastatic
Metastasis to other organs is the major cause of cancer-related deaths, a topic that is still quite a biological mystery. At Baylor College of Medicine, the lab of Dr. Xiang H.-F. Zhang has been investigating this phenomenon for many years and contributing breakthrough discoveries that provide answers to some of the intriguing questions surrounding metastasis.
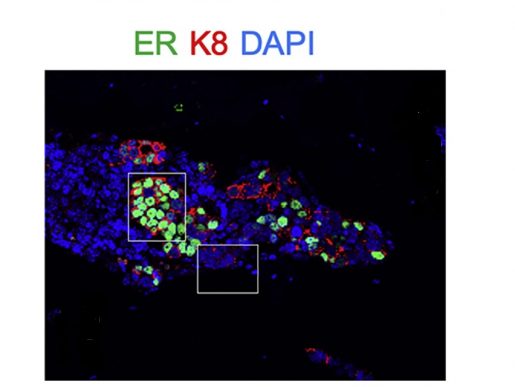
“Breast cancer mostly metastasizes to the bone; however, we still don’t know why, in more than two-thirds of cases, metastases will not be limited to the bone, but rather subsequently occur in other organs and eventually cause death,” said Zhang, William T. Butler, M.D., Endowed Chair for Distinguished Faculty and professor of molecular and cellular biology at Baylor. “Specifically, we have been studying why estrogen receptor-positive (ER+) breast cancer sometimes grows back in the bone and spreads to other tissues despite effective endocrine therapies directed at ER.”

In two papers published simultaneously, Zhang and his colleagues applied a series of models and techniques they had previously developed to investigate cancer-bone interactions at a single cell resolution to see what happens to ER+ breast cancer cells when they metastasize to the bone. They wanted to find out what might contribute to their resistance to endocrine treatment and enhance metastasis to other organs.
The bone microenvironment plays a role in metastasis
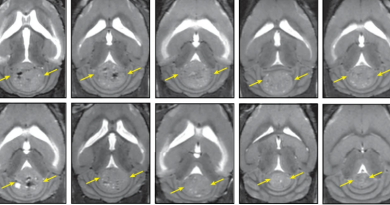
“Surprisingly, we discovered that when ER+ breast cancer cells locate in the bone, they reduce their expression of ER, which makes them less susceptible to endocrine therapies directed at ER,” said co-first author Dr. Igor Bado, a postdoctoral fellow in the Zhang lab. “We determined that osteogenic cells, the cells that make new bone, promoted this change in the cancer cells both by releasing factors and by direct physical interaction with the cells.”

Interestingly, the interaction with osteogenic cells also triggered changes in gene expression that gave the cancer cells stem cell-like properties, such as unchecked self-renewal and differentiation into various cell types. Having these properties, the authors explain, makes cancer cells more capable of generating new metastasis.
The team identified a number of metabolic pathways that were altered in cancer cells by the bone microenvironment. “Among these pathways, the EZH2-mediated pathway drives ER+ breast cancer cells toward a stem-like state. Inhibiting EZH2 reversed endocrine resistance,” Bado said.
“EZH2 is emerging as a leading candidate for therapeutic intervention,” said Zhang, a McNair Scholar and member of Baylor’s Dan L Duncan Comprehensive Cancer Center.
These findings were published in Developmental Cell.
‘Empowering’ cancer cells to spread further
These findings readily connected with the work Dr. Weijie Zhang was conducting in the Zhang lab. “We were studying whether bone metastases, as compared to a primary tumor, were more likely to further disseminate to other organs,” said Weijie Zhang, co-first author of the work and a postdoctoral fellow in the Xiang Zhang lab. “We found that the bone microenvironment is like a ‘powering station’ for cancer cells, enhancing their ability to further disseminate to other organs. Our findings support the idea that many metastases may be initiated, not by primary tumors, but by further spread of other metastases.”

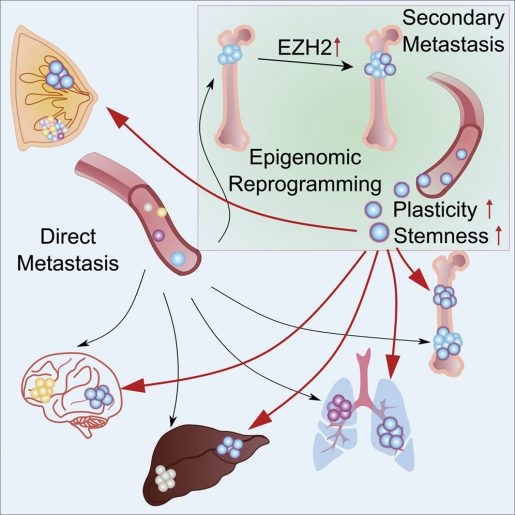
The researchers also showed that the bone microenvironment can empower other types of cancer, such as prostate cancer.
“This is something that other people have not observed before,” Weijie Zhang said. “We were able to discover this thanks to our unique model in which we can confine cancer cells to the bone to start with, which allows us to follow subsequent dissemination. We published this discovery in the journal Cell.”
“Taken together, these studies revealed an unappreciated role of the bone microenvironment in metastasis progress and elucidated a reprogramming process driving terminal-stage, multi-organ metastases that provides new insight into the clinical enigma of ER+ metastatic recurrences despite endocrine therapies,” Xiang Zhang said.
For the complete list of contributors and financial support for the study published in Developmental Cell, follow this link. Click here to find the contributors and financial support for the work in Cell.