Using proteogenomics to improve the treatment of squamous cell carcinoma
Patients with head and neck squamous cell carcinoma (HNSCC), the sixth most common epithelial cancer worldwide, are treated with surgery, chemotherapy and radiotherapy. In addition, targeted agents, including an EGFR monoclonal antibody (mAb) inhibitor and two programmed cell death protein 1 (PD-1) inhibitors, have been approved by the U.S. Food and Drug Administration for HNSCC treatment, but response rates are moderate.
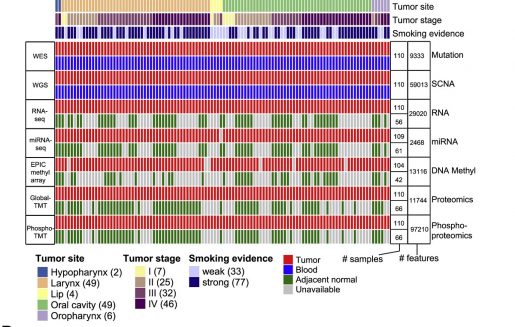
In this study, researchers led by Baylor College of Medicine, Johns Hopkins University and the National Cancer Institute’s Clinical Proteomic Tumor Analysis Consortium (CPTAC) investigated what new insight proteogenomic analysis might offer into understanding why certain patients respond to certain treatments while other patients do not. They propose that their findings may help better match patients to an appropriate course of treatment in the future.
Building a proteogenomic profile
The team profiled proteins, phosphosites (a site on a protein associated with phosphorylation) and signaling pathways in 108 human papillomavirus-negative HNSCC tumors in order to understand how genetic aberrations drive tumor behavior and response to therapies.
“We found three subtypes of head and neck squamous cell carcinoma, and each subtype may be a good candidate for a different type of therapy – EGFR inhibitors, CDK inhibitors or immunotherapy,” said Dr. Bing Zhang, lead contact of the study and professor in the Lester and Sue Smith Breast Center and the Department of Molecular and Human Genetics at Baylor. “We also identified candidate biomarkers that could be used to match patients to effective therapies or clinical trials.”

Finding effective biomarkers
One important finding involved matching HNSCC patients to EGFR mAb inhibitors. Cetuximab, an EGFR mAb medication, was approved by the FDA in 2006 as the first targeted therapy for HNSCC, however the success rate for this treatment is low. Moreover, EGFR amplification or overexpression cannot predict response to EGFR mAbs. In this study, researchers found that EGFR ligands, instead of EGFR itself, act as the limiting factor for EGFR pathway activation. When ligand is low, the downstream pathway will not be triggered, even if EGFR protein is highly overexpressed.
We proposed that the EGFR ligand should be used as a biomarker, rather than EGFR amplification or overexpression, to help select patients for the EGFR monoclonal antibody treatment,” said Zhang, a member of the Dan L Duncan Comprehensive Cancer Center, a Cancer Prevention & Research Institute of Texas (CPRIT) Scholar and a McNair Scholar at Baylor.
“Tumors with high EGFR amplification do not necessarily have high levels of EGFR ligands, which may underlie their lack of response to EGFR mAb therapy.” The team confirmed this hypothesis by analyzing previously published data from patient-derived xenograft models and a clinical trial.
Additionally, tracking a key tumor suppressor known as Rb (retinoblastoma), the research team identified a striking finding that suggests that Rb phosphorylation status could potentially be a better indicator of a patient’s response to CDK4/6 inhibitor therapy. The study showed that the many mutations in the genes regulating CDK4/6 activity were neither necessary nor sufficient for activation of CDK4/6.
The team found that the CDK4 activity was best measured through Rb phosphorylation measurements, thus identifying a potential measure for patient selection in CDK inhibitor clinical trials.
Immunotherapy insights
The research team also found important insights into the effectiveness of immunotherapy. PD-1 inhibitors target the interaction between immune checkpoints PD-1 and PD-L1, but success rates of immunotherapy are low, even when PD-L1 expression is used for patient selection. The researchers examined tumors with high expression of PD-L1 and found that when a tumor overexpresses PD-L1, it also upregulates other immune checkpoints, thus allowing the tumor growth despite the use of PD-1 inhibitors.
This observation suggests that PD-1- and PD-L1-activated tumors with ‘hot’ immune environments may require multiple types of immunotherapy, which target different immune checkpoint proteins, to be effective. Conversely, tumors with ‘cold’ immune environments are not good targets for immunotherapy.
‘Immune-cold tumors’ are tumors that contain few if any infiltrating immune T cells. Examination of how a tumor becomes immune-cold showed that the problem stems from a flaw in its antigen presentation pathway, a first step toward triggering an immune response against tumor antigens. In immune-cold tumors multiple key gene components of the antigen presentation pathway were deleted. As a result, although tumor antigens are being expressed, the immune system is not able to recognize them on the surface of cancer cells and therefore fails to activate the body’s defense system against the tumor. These deletions have the potential to be effective targets for future therapies.

“This study extends our biological understanding of HPV-negative HNSCCs and generates therapeutic hypotheses that may serve as the basis for future studies and clinical trials toward molecularly-guided precision medicine treatment of this aggressive cancer type,” said Dr. Daniel W. Chan, co-corresponding author of the study, professor of pathology and oncology, and director of the Center for Biomarker Discovery and Translation at the Johns Hopkins University School of Medicine.
Find all the details of this study and a full list of contributing authors in the journal Cancer Cell.
This work was supported by grants U24 CA210954, U24 CA210985, U24 CA210972, U24 CA210979, U24 CA210986, U24 CA214125, U24 CA210967, and U24 CA210993 from the National Cancer Institute (NCI) Clinical Proteomic Tumor Analysis Consortium (CPTAC), by a Cancer Prevention Institute of Texas (CPRIT) award RR160027, by grant T32 CA203690 from the Translational Breast Cancer Research Training Program, and by funding from the McNair Medical Institute at the Robert and Janice McNair Foundation.
By Molly Chiu