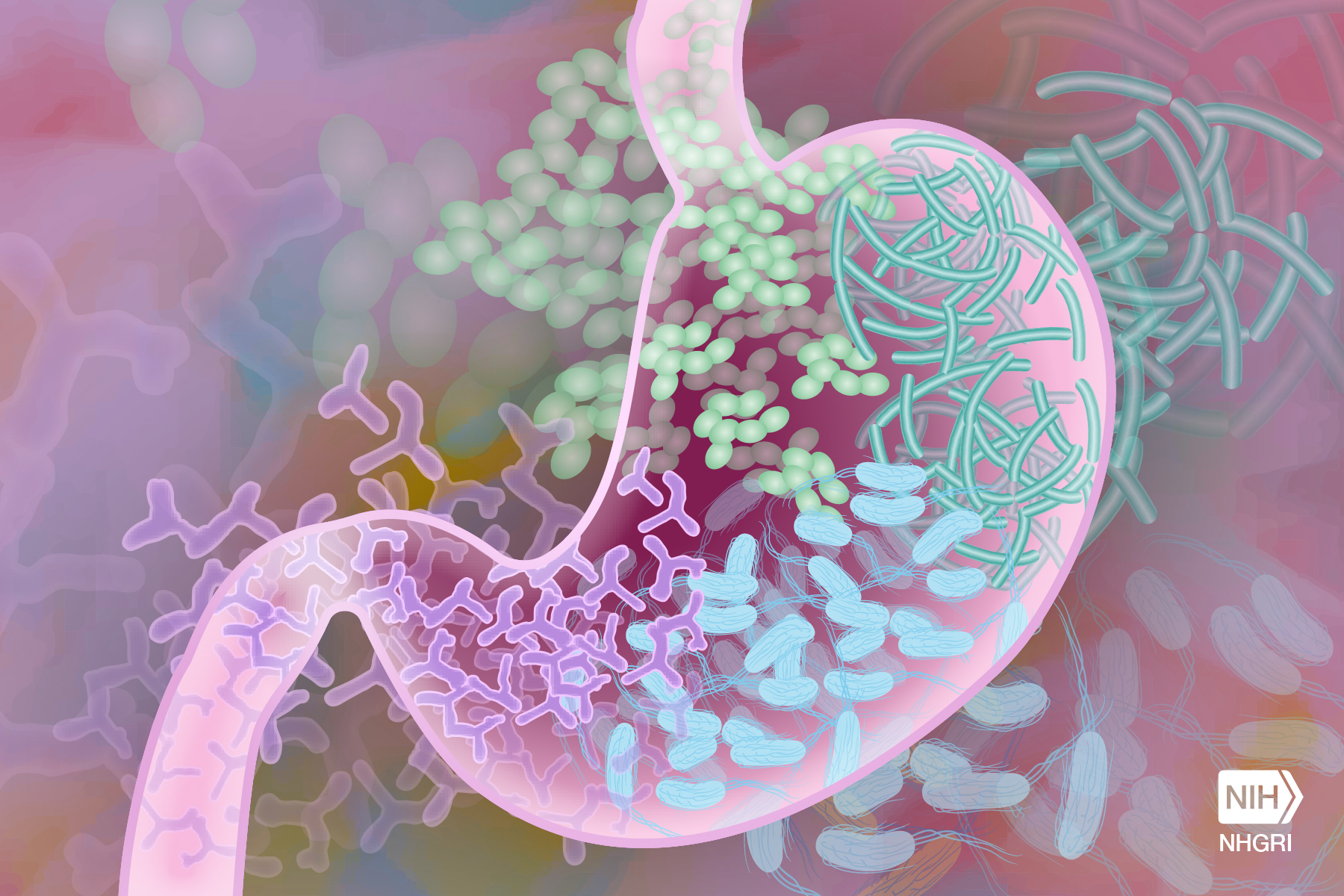
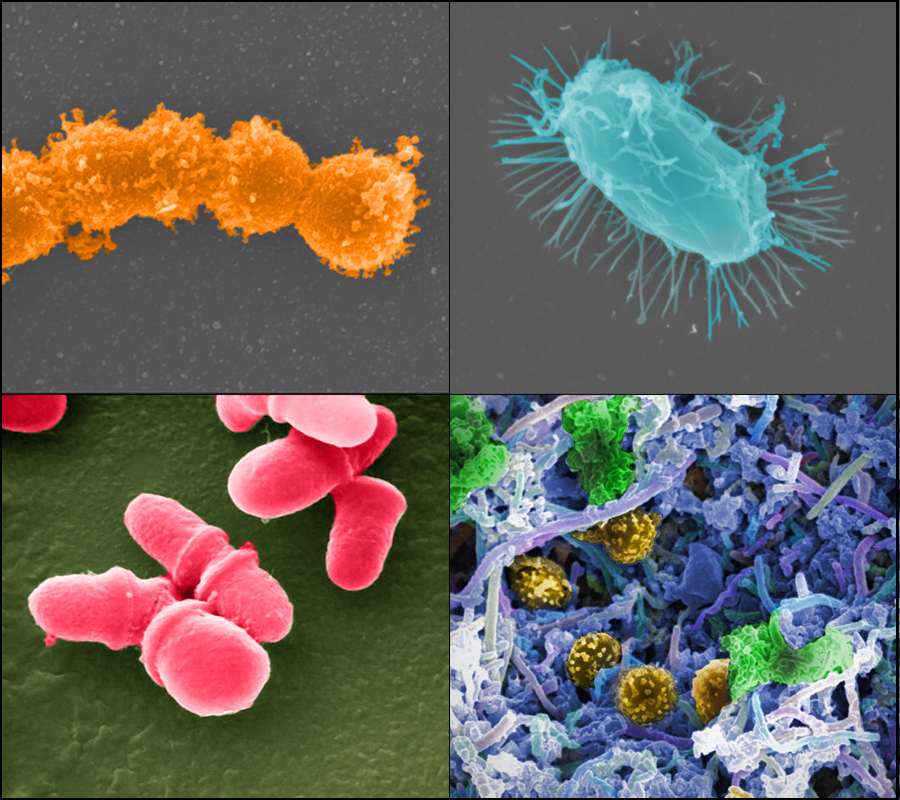
Gut metabolite profile may provide insight into how NEC happens
Necrotizing enterocolitis (NEC) is a serious disease mediated by an inflammatory process that leads to intestinal damage and sometimes death. It is rare in full term babies, but it occurs more often in preterm infants. The earlier before full term the infant is born, the higher the risk of developing the disease.
The risk of preterm infants developing the disease also is higher when they are fed formula than when they feed on breast milk. Of the many reasons why breast milk protects preterm infants from this serious condition better than formula, not all of them are well understood.
“Although this condition has been known for more than 50 years, we still don’t understand exactly how it develops. In this study we worked with a premature piglet model of NEC that closely reproduces the characteristics observed in human infants,” said Dr. Douglas Burrin, research physiologist at the USDA-ARS Children’s Nutrition Research Center and professor of pediatrics at Baylor College of Medicine.

The researchers had previously shown that the type of carbohydrate or sugar in the diet of preterm piglets affected the risk of developing NEC. Here, they investigated whether there was a connection between the type of carbohydrates in formula; the products of metabolism or metabolites; and the risk of NEC.
Preterm infant formulas contain two main sugars, lactose and corn syrup solids. The main carbohydrates in human milk are lactose and oligosaccharides. Burrin and his colleagues had shown that feeding formula containing the sugar lactose protected preterm piglets from NEC. In comparison, piglets fed formula containing corn syrup solids had a higher risk of developing the condition.
In this study, the researchers took a closer look at the effect of these two different sugars on the development of NEC using a more detailed analysis than had been done in previous studies: they characterized the bacterial communities, or microbiome, of the gut, and the metabolite profiles found in the gut and the blood.
“We asked, how does diet interact with the microbiome in the developing piglet gut, and how does that interaction shape not only the community of bacteria but also the metabolites found in the gut and blood of the piglets,” Burrin said.
To identify the metabolites produced by the piglets, Burrin and his colleagues partnered with Metabolon, a company that specializes in the analysis of all the main products of metabolism and their intermediates in a cell, tissue or organism. They also partnered with the Alkek Center for Metagenomics and Microbiome Research at Baylor College of Medicine to analyze the microbiome in the piglets, comparing the effects the two diets have on the profiles of bacteria growing in their gut.
The researchers confirmed that formula with lactose can protect piglets from developing NEC better than formula with corn syrup solids. Fourteen percent of the piglets in the lactose-containing formula developed NEC, while 44 percent of the piglets in the corn syrup solids-containing formula presented with the condition.
We determined that the communities of bacteria were not dramatically different between the lactose and the corn syrup solids group, but the metabolite profiles were quite different,” said Burrin.
“We found a clear association between the metabolite profile, the sugar in the diet and the risk of developing NEC. This suggested that the metabolite profile is perhaps an early determinant of the development of this disease, as the microbiome begins to develop,” Burrin said.
“We think that the metabolite profiles we identified correlating with disease are going to be useful for generating and testing new hypotheses to better understand how this disease happens,” said first author Lee Call, a graduate student in the Burrin lab.

The researchers’ next plan is to study in more detail what these metabolites that correlate with the disease are doing to lead to the condition, for instance, what biological pathways they are triggering or how they interact with cells in the gut.
Read the complete study in the journal Microbiome.
Other contributors to this work include Barbara Stoll and Berthe Oosterloo from USDA-ARS Children’s Nutrition Research Center, Baylor College of Medicine; Nadim Ajami, Joseph Petrosino, Fariha Sheikh and Oluyinka Olutoye from Baylor College of Medicine; and Anja Wittke, Rosaline Waworuntu and Brian Berg from Mead Johnson Pediatric Nutrition Institute
This work was supported in part by federal funds from the USDA, Agricultural Research Service under Cooperative Agreement Number 3092-51000-060-01, grants from Mead Johnson Nutrition, the Alkek Center for Metagenomics and Microbiome Research (CMMR) and the Texas Medical Center Digestive Diseases Center (NIH Grant P30 DK-56338). Further support was provided by the National Institutes of Health Grant T32-GM088129 and Gulf Coast Consortia, NLM Training Program in Biomedical Informatics (T15-LM007093).