Interferon drives cognitive impairment in Alzheimer’s disease model
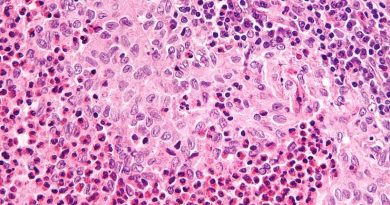
Alzheimer’s disease (AD), the most common type of dementia, progressively impairs memory, concentration and the ability to learn new things and accomplish everyday activities. Although scientists do not yet fully understand the causes of cognitive impairment associated with AD, the lab of Dr. Wei Cao and colleagues at Baylor College of Medicine have moved this understanding forward with the discovery of a major driver of memory and cognitive loss in a mouse model of the disease. Important because of its potential clinical implications, blocking this process reversed these memory and cognitive deficits in the animal model.
“The current understanding is that, in addition to having β-amyloid plaques and tau protein tangles, the brains of patients with Alzheimer’s disease have a marked inflammatory response, which might be more of a problem than plaques themselves,” said Cao. She was an associate professor of molecular and human genetics and the Huffington Center on Aging at Baylor during the development of this project. She is currently a professor of anesthesiology at UT Health-Houston.

IFN drives inflammation in brains with Alzheimer’s disease
In their previous study, Cao and her colleagues uncovered evidence that certain kinds of β-amyloid plaques activate microglia, resident immune cells of the brain, which then produce type I interferon (IFN). That in turn triggers a cascade of inflammatory reactions that lead to the loss of synapses, the junctions between neurons through which they communicate. Synapse loss is a key part of neurodegeneration and can lead to memory loss and eventually dementia. They found evidence of this crucial role of IFN both in animal models and in samples from patients with Alzheimer’s disease.
In the current study, the researchers worked with an animal model of Alzheimer’s disease, specifically with old mice that present with significant disease progression, including memory and cognitive decline when tested in memory assays. The researchers investigated the effect of IFN on memory and cognition using different approaches such as therapeutic antibody blockade and genetic knockouts.
“We were intrigued by the striking findings from these AD mice,” Cao said.
We found that the IFN-mediated inflammation pathway is rather harmful to the synapses, affecting memory and cognitive performance, and that, importantly, blocking the pathway restores these cognitive abilities.”
Cognitive impairment in AD requires concerted actions from different cells

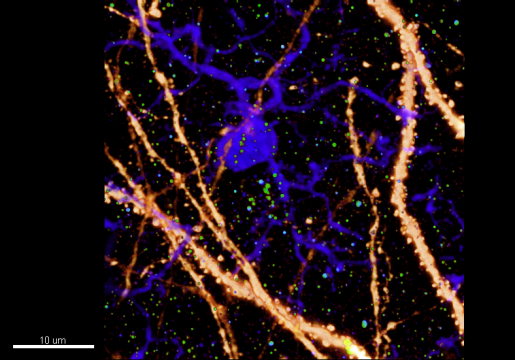
Interestingly, the team also found that certain, but not all, microglia were activated by IFN, as were other cells in the brain, such as neurons, astrocytes and blood vessels. “Our findings point to another layer of the complexity of IFN-mediated activation under AD conditions that hasn’t been fully appreciated,” explained first author, Dr. Ethan Roy, a postdoctoral researcher in the Cao lab.
“Another novel aspect of this work is that, using genetic strategies, we have found different functions of IFN signaling for different types of cells in the brain,” Roy said. “The full mechanism of synaptic loss requires concerted actions from different cells.”
The group also detected less amyloid plaques when neural cells were unable to respond to IFN. “Amyloid plaques trigger IFN production, then IFN activation promotes more plaque formation. Unfortunately, this feedforward loop is operating in AD,” Cao said.
The researchers are now interested in investigating how IFN signaling affects later stages of AD development. “We are exploring developing new therapeutic strategies that target IFN pathways to slow down cognitive decline in mice and, hopefully, eventually in people,” Cao said.
Read the complete study in the journal Immunity.
Other contributors to this work include Gabriel Chiu, Sanming Li, Nicholas E. Propson, Rupa Kanchi, Baiping Wang, Cristian Coarfa and Hui Zheng, all at Baylor College of Medicine.
The study was funded by National Institutes of Health grants (AG057587, AG020670, AG062257 and NS093652), BrightFocus (ADR A20183775), Brown Foundation 2020 Healthy Aging Initiative and Cure Alzheimer’s Fund. Further support was provided by IDDRC grant U54HD083092 from the Eunice Kennedy Shriver National Institute of Child Health & Human Development.