It’s all in the delivery – Antibodies improve nanoparticle delivery of therapeutic nucleic acids
Research has shown that some nucleic acids, such as messenger RNA (mRNA), DNA and related molecules, can be effective disease treatments in laboratory settings, raising expectations that improved human therapies could be developed for conditions including cancer and genetic and vascular diseases. Therapeutic nucleic acids (TNA) can potentially serve as tools, drugs or vaccines for the treatment, prevention or diagnosis of conditions at the genetic level.
“Currently, a handful of TNAs have been approved for clinical applications and clinical trials are on their way” said Dr. Changyi Johnny Chen, professor of surgery and molecular and cellular biology at Baylor College of Medicine. “However, progress is limited mainly because TNAs typically are difficult to deliver effectively in living organisms.”

When naked nucleic acids enter the body, they encounter several obstacles that limit their availability. For instance, they can face endothelial barriers that reduce their desired distribution. They also can confront quick enzymatic digestion, liver retention, renal clearance and unexpected accumulation in the ‘wrong’ tissue. TNAs can activate the immune system and cause severe side effects.
“Despite the obstacles limiting their applications, TNAs are promising approaches to treat a variety of conditions. My lab is interested in developing strategies that can increase their safety and efficacy by improving their delivery,” Chen said.
One of the most widely used delivery systems is liposomes, artificially formed tiny spherical sacs of lipids enclosing a water droplet that carries TNAs. Virus-based delivery systems also have been used. Both strategies have been useful, but pose efficacy and safety challenges.
LGA-PEI nanoparticles

Since they moved to Baylor in 2002, the lab of Dr. Qizhi Cathy Yao, professor of surgery, molecular virology and microbiology and pathology and immunology, and Chen’s lab have been working on developing effective therapeutics for pancreatic cancer.
“Our previous findings have shown that certain RNAs, such as microRNA-198 (miR-198), a natural tumor suppressor, could have clinical applications in cancer,” Chen said. “If they did, we were going to need an effective and safe delivery system, which was not available at the time.”
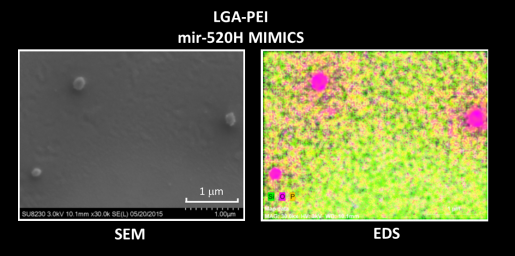
“For many years, my laboratory has been developing TNA delivery systems,” Chen said. “Finally, we designed, synthesized and characterized a new approach based on polyethylenimine (PEI) polymers modified with lactic-co-glycolic acid (LGA), called LGA-PEI.”
LGA-PEI polymers spontaneously form nanoparticles with layers that envelope the TNAs such as miR-198, protecting them from enzymatic digestion and enhancing cellular uptake.
For cancer, enhanced uptake of TNA-loaded nanoparticles could lead to improved anti-cancer effects. Experiments with cells in the lab and mouse models showed that LGA-PEI nanoparticle delivery of TNAs was less toxic than that of previous nanoparticles.
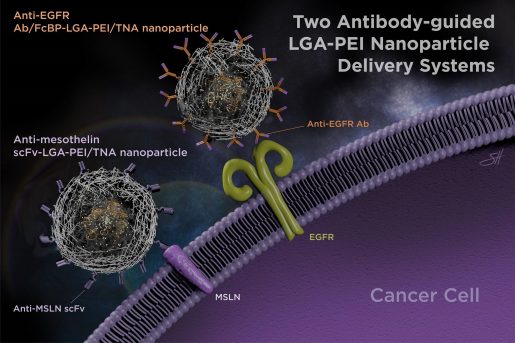
“Our results were encouraging, but we still wanted to improve our delivery system further,” Chen said. “In this study we added antibodies or antibody fragments to our LGA-PEI nanoparticles to fine tune their delivery to specific cancers.”
Antibodies improve delivery
Antibodies are proteins produced by the immune system that can recognize and bind to specific markers on the cell surface. When antibodies are attached to other molecules or to nanoparticles, they can mediate their delivery to specific cancer cells displaying unique markers that are not in other cells, targeting cancer cells while mostly sparing normal cells.
For example, Chen and others previously showed that the protein mesothelin (MSLN) is abundantly expressed in some human cancers, including pancreatic cancer. However, MSLN has limited expression in normal tissues of the body, which makes it a candidate for targeting cancer therapy.
“In the current study, we successfully linked a fragment antibody against MSLN to our LGA-PEI polymer and used fluorescent tags to show improved MSLN-targeted delivery of TNAs in pancreatic cancer cells grown in the lab and in animal models,” Chen said.
The Chen lab is interested in conducting studies that will test the effectiveness of this active-targeting delivery system at inhibiting tumor growth and metastasis as well as at improving the overall survival of tumor-bearing animals as compared with passive-targeting delivery or other non-targeting delivery systems.
The improved nanoparticle delivery system is a technological platform that can be extended to other cancer types or diseases for the development of new treatments, molecular diagnosis strategies or vaccines.
Find the complete study in the journal Pharmaceuticals.
Other contributors to this work include Jian-Ming Lü, Zhengdong Liang, Dongliang Liu and Bin Zhan. The authors are affiliated with Baylor College of Medicine and/or Michael E. DeBakey VA Medical Center.
This project has been partially supported by Baylor College of Medicine Alkek Award Pilot Projects in Experimental Therapeutics, Michael E. DeBakey Department of Surgery Seed Fund and National Institutes of Health R01 award CA183984.