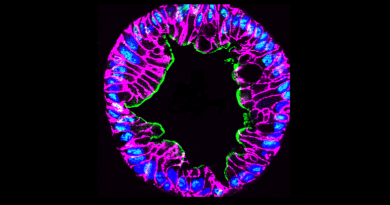
Uncovering a pathway that protects the uterus from cancer
The TGF-beta signaling pathway regulates the development of many types of cancer, but it was not clear whether it also played a role in uterine cancer, the most common gynecological cancer in the U.S. In his lab at Baylor College of Medicine, Dr. Martin M. Matzuk and his colleagues have explored the potential role of this pathway in uterine cancer by developing two new mouse models of the disease.
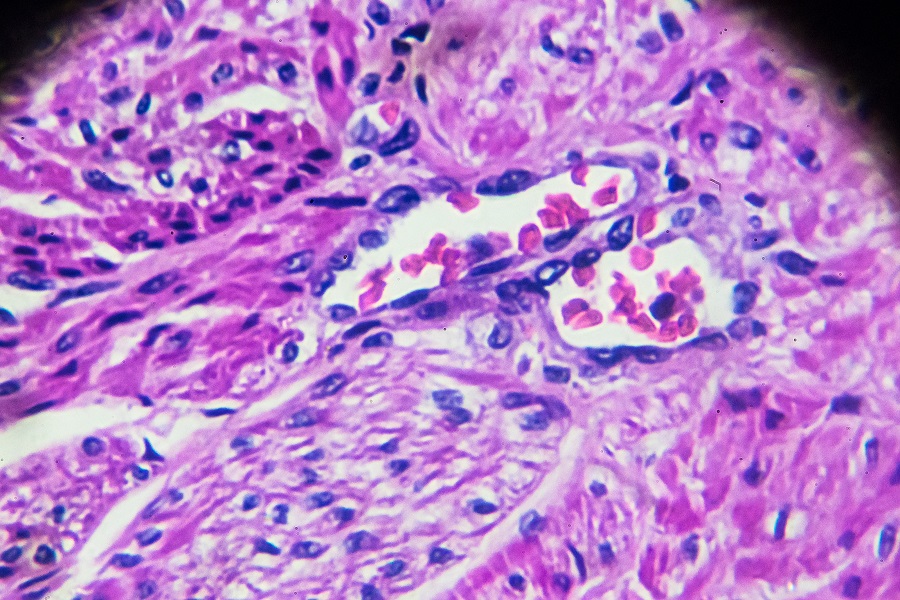

“Each mouse model tested the effect of lacking specific proteins participating in the TGF-beta pathway,” said Matzuk, who is director of the Center for Drug Discovery, Stuart A. Wallace Chair, Robert L. Moody, Sr. Chair and professor of pathology & immunology at Baylor College of Medicine. “One model was studied in the absence of the protein TGF-beta receptor ALK5 and the other model tested the inactivation of both Smad2 and Smad3, proteins downstream of the receptor.

To develop the first mouse model, Dr. Diana Monsivais, instructor of pathology & immunology at Baylor, Dr. Jia Peng, a former postdoctoral associate at Princeton University and their colleagues genetically modified mice to lack ALK5, which mediates TGF-beta signals to the cell. Then, they observed whether the absence of ALK5 led to the transformation of endometrial cells into cancer cells.
“The mice lacking ALK5 developed metastatic endometrial tumors with cervical and vaginal masses. The tumors were estrogen-dependent, meaning they required estrogen to grow. When the ovaries – the natural source of estrogen – were removed, both the tumors and metastases to the lungs regressed,” Monsivais said. “These and other results indicate that it is important to maintain TGF-beta signaling via ALK5 to sustain the health and normal function of the uterus.” This study is in this PNAS article.

The second mouse model was developed by Dr. Maya Kriseman, reproductive endocrinology and infertility fellow in the Department of Obstetrics and Gynecology at Baylor College of Medicine, and her colleagues. In this case, the researchers genetically modified mice to lack two other proteins, Smad2 and Smad3, which transmit the signal from the TGF-beta receptors to the nucleus. Then, they assessed the effect of lacking these proteins in tumor development.
“I found that disrupting these particular proteins, Smad2 and Smad3 in mice, caused a very aggressive hyperplasia or overgrowth of the endometrium that rapidly developed into cancer and death,” Kriseman said. “These effects were dependent on estrogen, which also is a characteristic of the most common type of uterine cancer in humans. These and other findings give us a better idea as to why these mice develop aggressive uterine tumors and escape normal endometrial regulation.” This study can be found in this PNAS paper.
Taken together, the findings of Monsivais and Kriseman strongly suggest that the TGF-beta pathway is a tumor-suppressor mechanism in the uterus,” said Matzuk, who also is a member of the Dan L Duncan Comprehensive Cancer Center at Baylor College of Medicine.
“We anticipate that the new mouse models we have generated can be further used to uncover other processes that can lead to uterine cancer.”
“Dissecting the complex cascade of biological events that are necessary for protecting the uterus from cancer and for sustaining normal fertility is critical. By revealing that the TGF-beta pathway plays a protective role against uterine cancer, this work has significantly advanced our understanding of the disease and opened novel avenues for future therapeutic strategies,” said Dr. William E. Gibbons, professor of obstetrics and gynecology and director of reproductive endocrinology and infertility at Baylor College of Medicine.
Financial support to Baylor College of Medicine for the two studies was provided by Eunice Kennedy Shriver National Institute of Child Health and Human Development grants R01-HD032067, R01-HD033438, and K99-HD096057 and the Institutional Research and Academic Career Development Award (IRACDA) K12-GM084897. Dr. Monsivais holds a Postdoctoral Enrichment Program Award from the Burroughs Wellcome Fund.
Other contributors to the work of Monsivais and Peng include Dr. Yibin Kang, affiliated with Princeton University and supported by the New Jersey Commission on Cancer Research Fellowship, the Brewster Foundation and National Institutes of Health grant R01-CA212410. Additional authors to the work of Kriseman and colleagues include Monsivais, Julio Agno, Ramya Masand and Chad Creighton, all at Baylor College of Medicine.