Mouse blood vessels and immune system can team up against cancer metastasis
Picture a shapeless, small mass of tumor cells inside an otherwise healthy organ. The tumor is growing, thanks, among other things, to its ability to trigger the formation of new blood vessels, a process called angiogenesis that supplies oxygen and nutrients to the cell mass. This discovery has led to therapies that aim at blocking angiogenesis to starve the tumor. The therapies can retard tumor growth; however, they can also have undesired effects, metastasis and resistance to treatments.
Dr. Xiang Zhang and his colleagues have been puzzled by this apparent contradiction, so they have taken a closer look at what goes on in the tumor microenvironment in breast cancer.
Mouse models have provided intriguing observations. Lin Tian, a graduate student in the Zhang lab, recalls that he began his project with several interesting clues.
“Some of the lab tumor mouse models with high potential for metastasis rarely formed spontaneous metastasis in immunocompetent mice. However, the tumors became metastatic in immunodeficient mice that lacked immune T cells,” Tian said. “How are the T cells preventing the spontaneous metastasis in immunocompetent mice?”


There were more intriguing observations. “If delivered directly into the circulation, these cancer cells were able to efficiently colonize distant organs, even in the presence of T cells,” Tian said. “Is the invasion of cancer cells into the blood limiting spontaneous metastasis? In addition, bioinformatics analysis showed that good prognosis markers, such as T cell response and low hypoxia, mostly appeared together, hinting at an underlying relationship between the immune system and blood vessels in the tumor.”
“The tumor environment includes tumor cells and other types of cells that are recruited to the tumor mass and help the tumor grow,” Zhang said. “Scientists have divided those cells into subpopulations and studied them separately. We think that for a more complete understanding of the entire tumor microenvironment we have to study it with all its cell types together.”
Blood vessels and the immune system talk to each other
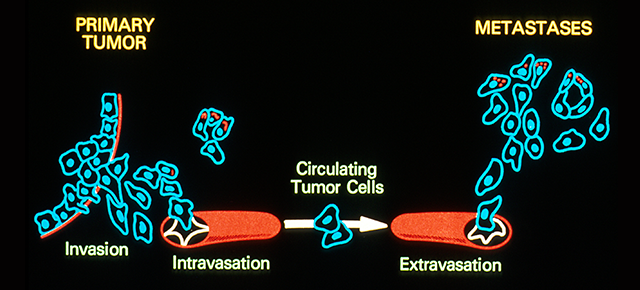
Despite their abundance, tumor blood vessels perform their job poorly because their structure and functions are abnormal. For instance, they leak, so they do not carry enough blood to the tumor which limits the amount of oxygen, known as hypoxia, and nutrients. This creates a harsh microenvironment that can stimulate some cancer cells to escape through gaps in blood vessels that can transport them to other organs. Abnormal tumor blood vessels may also interfere with anti-tumor immune cells or drugs attempting to reach the tumor.
Recently, restoring tumor blood vessels’ function and structure to normal, a process called vessel normalization, has been considered a promising strategy to improve anti-blood vessel therapies. Vessel normalization can potentially limit or prevent cancer progression and metastasis, and improve the response to immunotherapy, chemotherapy and radiation therapy. However, little is known about how the tumor microenvironment regulates vessel normalization.
By applying their approach of studying the tumor microenvironment as a whole, Zhang and his colleagues discovered that immune cells called T lymphocytes can promote the process of vessel normalization.
“The reciprocal regulation is also true,” Zhang said.
“If we change the structure of the vasculature of the tumor toward vessel normalization, then we can also stimulate T cells to infiltrate the tumor. This bidirectional regulation between blood vessels and the immune system had not been elucidated before.”
In this video, Dr. Xiang Zhang explains how this study could change the way researchers look at anti-cancer therapies.
Implications for cancer therapy
“Both anti-blood vessel therapies and immunotherapies have been used in the clinic and have different degrees of success,” said Zhang. “For instance, immunotherapy has been successful in some types of cancer such as melanoma and lung cancer, which are very aggressive cancers that did not have effective treatment until these therapies came along. However, there are still many patients who do not respond to this type of therapy. Similarly, from the anti-blood vessel therapy we know that many attempts have not been as successful as anticipated, and we have not been able to understand why.”
The researchers work suggests that therapies should consider not only, for instance, the presence of immune cells and their activities in the tumor microenvironment, but also the vascular structure of the tumor because it has implications of whether the tumor would be able to respond to immunotherapy.
“Our finding in mouse models suggests that anti-cancer therapies toward the blood vessels most likely influence the anti-tumor immune response, and vice-versa. Therefore, there is a possibility that we can achieve better outcomes by combining the therapies ,” Tian said.
“However, we are still far from having practical clinical solutions,” Zhang said. We hope that our work will provide some therapeutic theoretical basis for those researchers using the two different therapies to collaborate and look at each other’s biomarkers and therapeutic strategies.”
For all the details about this study, read the article in Nature.
Dr. Xiang Zhang is an associate professor of molecular and cellular biology in the Lester and Sue Smith Breast Center, and a McNair Scholar at Baylor College of Medicine.
###
Other contributors to this work include Amit Goldstein, Hai Wang, Hin Ching Lo, Ik Sun Kim, Thomas Welte, Kuanwei Sheng, Lacey E. Dobrolecki, Xiaomei Zhang, Nagireddy Putluri, Thuy L. Phung, Fabio Stossi, Arun Sreekumar, Michael A. Mancini, William K. Decker, Chenghang Zong and Michael T. Lewis, all from Baylor, and Sendura A. Mani at MD Anderson Cancer Center.
This work was supported by the Breast Cancer Research Foundation (NCI CA151293), the U.S. Department of Defense (DAMD W81XWH-16-1-0073, Era of Hope Scholarship; DAMD W81XWH-13-1-0296), Susan G. Komen (CCR14298445), and the McNair Medical Institute. Further support was provided by the Helis Foundation, the CPRIT Core Facility (Support Award RP120092), the National Cancer Institute (2P30CA125123-09), the Dan L. Duncan Comprehensive Cancer Center, the National Institutes of Health (P30 AI036211, P30 CA125123, 1S10OD016167 and S10 RR024574) and the National Science Foundation (DBI-0735191).