The hunger-satiety switch
When we are hungry, we eat; when we are full, we stop. Most of the time. Breaking this rule, however, can lead to overeating, obesity and diabetes or, the opposite, anorexia. The brain holds the secret to appetite control and it turns out that in mice a few thousand brain cells out of an estimated 75 million neurons in the mouse brain can control the animal’s feeding behavior.
This relatively small number of brain cells – the AgRP/NPY neurons located in the hypothalamus – is very important to the mouse.
“If the mouse does not have them, it will stop eating and die in a few days,” said Dr. Yong Xu, associate professor of pediatrics at the USDA/ARS Children’s Nutrition Research Center at Baylor College of Medicine and Texas Children’s Hospital and of molecular and cellular biology at Baylor.
Xu and colleagues have taken a closer look at how AgRP/NPY neurons regulate their electrical activity.

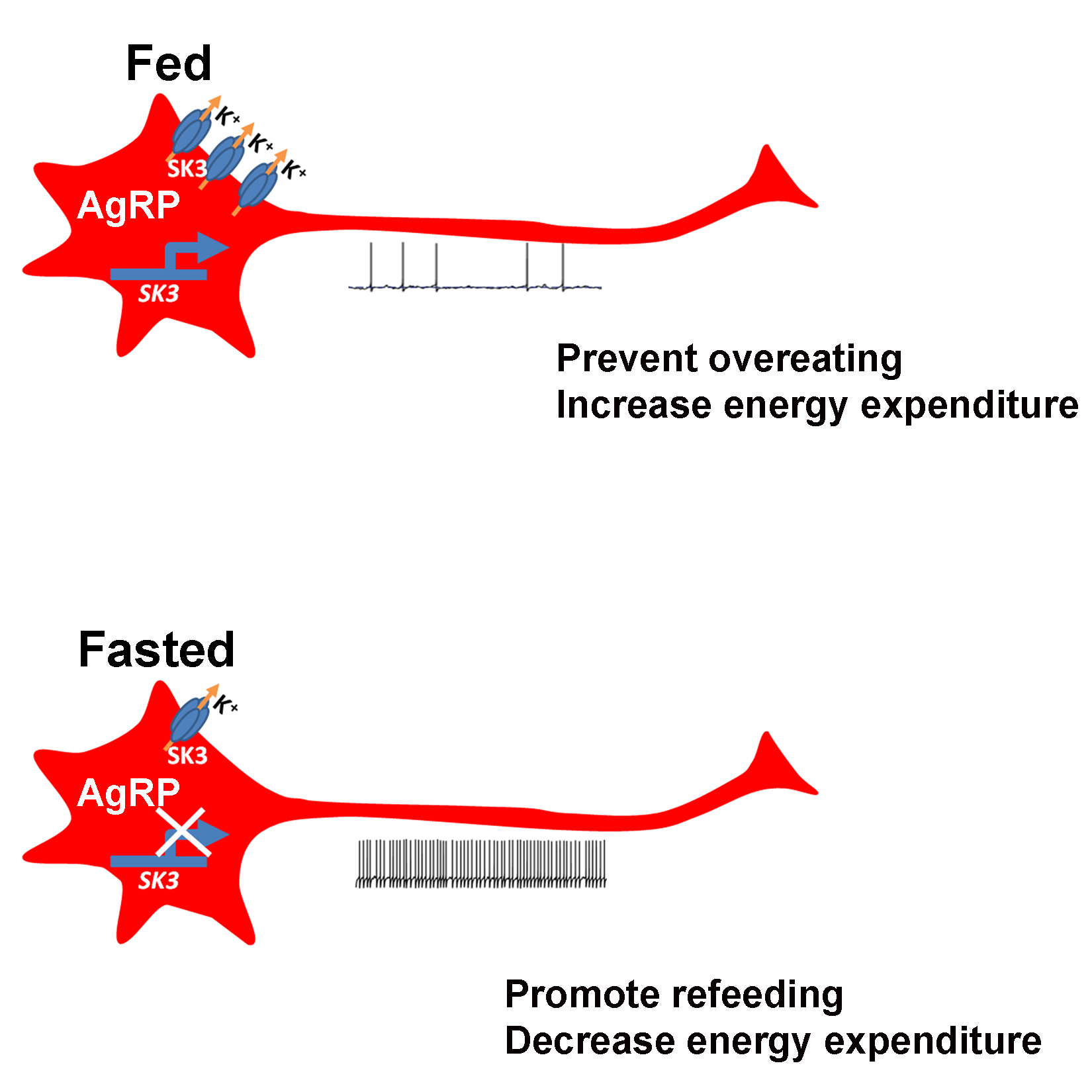
“The firing of electrical activity by AgRP/NPY neurons is tightly controlled by the nutritional status of the animal,” Xu said. “When the mouse is hungry the neurons fire more; when it is full, they fire less.”
The level of firing triggers the corresponding mouse behavior; when the neurons fire more, the mouse looks for food and shows less anxiety and aggression, which helps it search for food and feed. When the neurons fire less, which is triggered by the mouse being satiated, the animal stops eating and does not search for food. The cycle repeats when the satiated mouse is hungry again.
The scientists then set out to uncover the molecular mechanism that regulates this switch of firing activity of the AgRP/NPY neurons.
The hunger/satiety switch
“We found that the levels of a protein called SK3 can change dramatically in AgRP/NPY neurons,” said Xu. “When the animals are well fed, almost all the AgRP/NPY neurons express high levels of SK3. When the animals fast, the levels of SK3 are much lower. Further research showed that SK3 is involved in controlling the transport of potassium in and out of the cell, and this happens only in this group of neurons.”
The scientists confirmed the role of SK3 in the regulation of mouse feeding behavior by deleting the Sk3 gene in mouse models. “Without SK3, the hunger/satiety switch is lost; AgRP/NPY neurons fire constantly and the animals eat all the time and become obese,” said Xu.
With all this evidence SK3 became a target of much interest to the researchers.
“Our work opens the door to the possibility of treating obesity and eating disorders by developing drugs directed toward SK3,” said Xu.
In addition, Xu and colleagues think that these findings may lead to a better understanding of why some people do not control their feeding behavior, which leads to obesity and diabetes.
For more information on this work, read the paper in Cell Reports.
Other contributors to this work include Yanlin He, Gang Shu, Yongjie Yang, Pingwen Xu, Yan Xia, Chunmei Wang, Kenji Saito, Antentor Hinton Jr., Xiaofeng Yan, Chen Liu, Qi Wu and Qingchun Tong. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, South China Agricultural University, the University of Texas Southwestern Medical Center at Dallas and the University of Texas Health Science Center at Houston.
This work was supported by grants from the National Institutes of Health (R01DK093587, R01DK101379 and R01DK092605), the American Diabetes Association (#7-13-JF-61, #1-15-BS-184), the American Heart Association postdoctoral fellowship and the Davis Foundation.