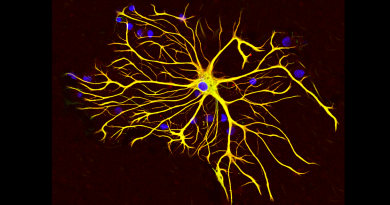
A new fruit fly model to probe an epileptic brain disorder
Developmental and epileptic encephalopathy (DEE) refers to a group of neurodevelopmental conditions characterized by developmental delay, cognitive impairment and seizures in children. In 2016, the first case linking variants in both the copies of UBA5 gene to DEE44 was reported. Since then, twelve distinct missense variants in the UBA5 gene have been identified in 25 patients.
Recently, Dr. Hugo J. Bellen and his team at Baylor College of Medicine and the Jan and Dan Duncan Neurological Research Institute (Duncan NRI) at Texas Children’s Hospital generated a new fruit fly model to assess the severity of symptoms caused by each of these variants. This systematic analysis lays the foundation for better evaluation of the variants, which is important for the DEE44 patients in the future and for the development of drugs and gene therapy to treat this rare disorder. The study was published in the journal eLife.
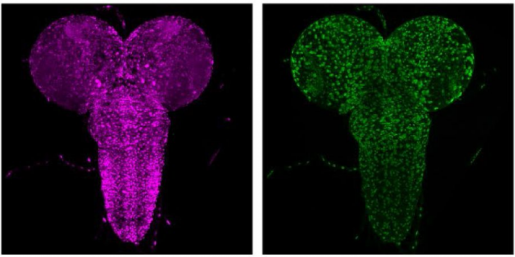
Understanding the UBA5 variants
The UBA5 gene is highly conserved across species ranging from fruit flies to humans. It encodes a protein that binds to Ubiquitin-fold Modifier 1 (UFM1) and was discovered two decades ago. It acts as an enzyme that catalyzes a modification called UFMylation. The exact mechanisms and biological significance of UFMylation are still unclear but it adds a new regulatory layer to many fundamental cellular processes. The fruit fly counterpart, Uba5, is expressed in a subset of neurons and glial cells in the fly’s central nervous system.
“We generated a humanized model by expressing the human version of the UBA5 gene in fruit flies lacking their own Uba5 gene,” said Dr. Xueyang Pan, the first author of the study and a postdoctoral fellow in the Bellen lab. “Next, we characterized the strength of the disease-causing variants by expressing them individually in flies and observing the changes they caused.”

They found that the severity and the type of symptoms varied widely among different variants. For instance, four variants failed to prevent death caused by the loss of endogenous Uba5. Another five variants caused progressive motor defects, three of which also caused developmental delays or seizure-like symptoms. While the normal human UBA5 substituted all essential functions of its fly counterpart, the disease-causing variants exhibited variable loss of function that was classified as mild, intermediate or severe. Importantly, overexpression of the UBA5 gene did not cause any obvious defects, suggesting a potentially safe therapeutic strategy to increase UBA5 enzyme levels, which is encouraging.
In collaboration with Dr. Jonathan Pruneda and Dr. Ruth Napier at Oregon Health & Science University and the Raiden Science Foundation of Beaverton, Oregon, the team also established several new biochemical assays to determine the impact of these disease-causing gene variants on the stability and activity of the UBA5 enzyme. The researchers found a close correlation between the severity of the symptoms exhibited by a particular variant in fruit flies and its enzymatic stability and/or function, as measured by biochemical assays. These findings provide a molecular explanation for the loss of function among UBA5 variants, and importantly establish a route to develop new drugs that restore UBA5 function and treat the disease.

“We undertook this study because there was a clear need to assess the disease-associated variants of UBA5,” said Bellen, Distinguished Service Professor of Molecular and Human Genetics at Baylor. “This study not only established a powerful experimental model to better understand how DEE44 develops, but also can serve as a template to better understand other human disorders and to develop new therapies for DEE44 and other conditions.”
Others involved in the study were Albert N. Alvarez, Mengqi Ma, Shenzhao Lu, Michael W. Crawford, Lauren C. Briere, Oguz Kanca, Shinya Yamamoto, David A. Sweetser and Jenny L. Wilson. Their institutional affiliations can be found here.
The study was supported by grants from the National Institutes of Health, the Department of Veterans Affairs, the Huffington Foundation, the Jan & Dan Duncan Neurological Research Institute at Texas Children’s Hospital, the OHSU Molecular Microbiology and Immunology Interdisciplinary Pilot Award, the Oregon Clinical and Translational Research Institute’s Biomedical Innovation Program NCAT from NIH, Baylor College of Medicine’s IDDRC, the Hill Family Fund for the Diagnosis, Management of Rare and Undiagnosed Diseases at Mass General, and the American Institute for Neuro Integrative Development Inc (AIND).
The original article by Rajalaxmi Natarajan, Ph.D. is posted here.
Follow From the Labs on Twitter @BCMFromtheLabs.