Researchers reveal genetic basis of severe forms of EBF3-associated neurodevelopmental disorders
The EBF3 gene encodes an evolutionarily conserved protein — a transcription factor — that regulates multiple neurological and non-neurological developmental processes. In 2016, Dr. Hsiao-Tuan Chao at Baylor College of Medicine and the Jan and Dan Duncan Neurological Research Institute (Duncan NRI) at Texas Children’s Hospital, and her colleagues discovered that loss of one functional copy of the EBF3 gene causes the disorders known as 10q26-deletion syndrome and hypotonia, ataxia and delayed development syndrome (HADDS).


The condition has presented quite a challenge to physicians and researchers as its presentation can vary widely among patients. In this study, Chao, assistant professor of pediatrics – neurology and of neuroscience and a McNair Scholar at the McNair Medical Institute at The Robert and Janice McNair Foundation at Baylor, and other researchers from the Undiagnosed Diseases Network and the Duncan NRI set out to better understand this condition.
The road to discovery
Shortly after Chao and her colleagues discovered the link between EBF3 and HADDS, she opened the world’s first EBF3-disorders-focused clinic at Texas Children’s and, along with Dr. Michael Wangler, assistant professor of molecular and human genetics at Baylor and Texas Children’s, evaluated the largest number of individuals with this condition at a single institution.

As Chao and her team evaluated more individuals with EBF3-related disorders, they were puzzled by the fact that some showed subtle symptoms, while others struggled with severe motor limitations and simultaneous neuropsychiatric conditions such as anxiety, autism or attention deficit hyperactivity disorder (ADHD).
EBF3-related disorders were identified just five years ago, so the full spectrum of symptoms and molecular links between the symptom profile and disease severity had not been explored prior to this study.
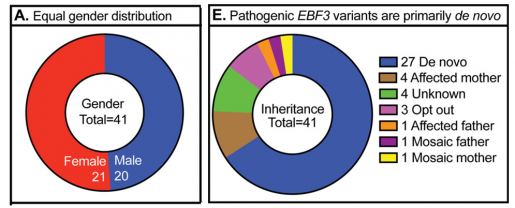
By combining a comprehensive meta-analysis of the medical literature with an extensive study of both EBF3 gene variants and patient characteristics, Chao and her team discovered that patients with EBF3 gene variants affecting a critical region — a zinc finger domain (ZNF) of EBF3 — suffer from a more severe form of EBF3-related autism spectrum and neurodevelopmental disorders than individuals with other variants.
“By systematically collating clinical information from the patients diagnosed with HADDS and using computational methods to uncover underlying associations between the precise nature and molecular location of the mutations with the severity of the disease symptoms, our team has come up with a refined understanding of this condition.
These findings enable physicians to predict the clinical course of the disease for each patient,” said Chao, faculty in the Department of Molecular and Human Genetics at Baylor.
“The comprehensive characterization of the spectrum of patient characteristics combined with algorithmic prediction and functional validation in fruit fly and cell culture assays was instrumental in making these discoveries,” said first author Cole Deisseroth, a medical student in the Chao lab who is enrolled in Baylor’s dual-degree Medical Scientist Training program. “Together, these approaches revealed the critical role of the ZNF in EBF3’s regulation of developmental processes and the relationship to disease severity.”

The findings of this study will help clinicians in predicting the potential trajectory of the disease and prognosis for each patient diagnosed with EBF3-related autism spectrum and related neurodevelopmental disorders. They also will provide the foundation for future mechanistic studies on EBF3-related disorders and will facilitate the development of targeted and personalized therapies for different sub-populations of affected individuals based on molecular findings.
“The quantitative analysis of patient characteristics and disease severity assessment algorithms developed in this study have the potential for broad applicability across many other human neurodevelopmental disorders, including autism spectrum disorders, and can lead to better diagnostic and prognostic predictions as well as new targeted therapies for those conditions,” Chao said.
Interested in all the details of this study? Find it in the Annals of Neurology.
Others involved in the study were Vanesa Lerma, Christina Magyar, Jess Pfliger, Aarushi Nayak, Nathan Bliss, Ashley Lemaire, Vinodh Narayanan, Christopher Balak, Ginevra Zanni, Maria Enza Valente, Enrico Bertini and Paul Benke. They are affiliated with one or more of the following institutions: Baylor College of Medicine, Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital, Case Western Reserve University, Texas A&M University, University of California San Diego, University of Pavia, Bambino Gesu Children’s Hospital, Joe DiMaggio Children’s Hospital and McNair Medical Institute.
The study was supported by the National Institutes of Health, the McNair Medical Institute at The Robert and Janice McNair Foundation, Burroughs Wellcome Fund, Mark A. Wallace Endowment and the Child Neurology Society and Foundation. Support also was provided by Baylor College of Medicine Medical Scientist Training Program, the 2021 Child Neurology Foundation Neurodevelopmental Disabilities Summer Research Scholarship, the Gordon and Mary Cain Foundation and Annie and Bob Graham, Autism Science Foundation, the European Reference Network for Rare Neurological Diseases and the Italian Ministry of Health.