Gene signature may give an upper hand in metastatic breast cancer
Identifying tumor-specific clues of potential poor outcomes could one day lead to improved treatments for metastatic breast cancer. At Baylor College of Medicine, Dr. Matthew J. Ellis and his colleagues are gaining ground in that direction with the discovery of a genetic signature that can provide specific details about a tumor that could guide physicians in the selection of more effective treatments.
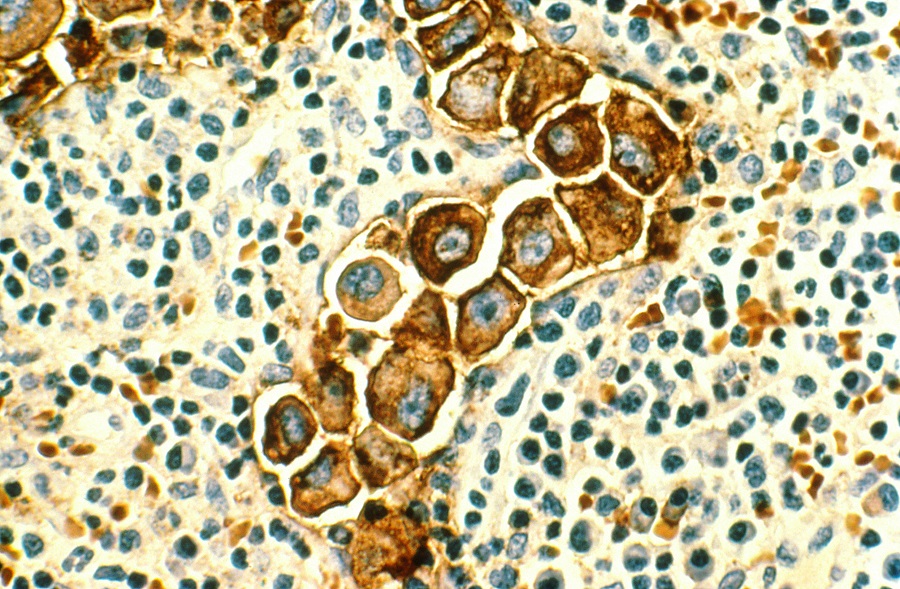
“About 80% of all breast cancers depend on the hormone estrogen to grow. The hormone promotes tumor growth by binding to the estrogen receptor (ER),” said Ellis, professor and director of the Lester and Sue Smith Breast Center and a McNair scholar at Baylor.

Disrupting the estrogen-ER interaction is a key therapeutic approach. Drugs such as tamoxifen and fulvestrant target the ER this way, but tumor cells learn to evade this attack and become resistant to these drugs.
“One of the predominant ways ER+ breast cancer cells evade treatment is by creating mutant ERs that no longer can be recognized and targeted by ER-targeting cancer drugs and therefore are unsusceptible to current therapies directed at disrupting estrogen-fueled tumor growth,” said first author Xuxu Gou, a graduate student in the Ellis lab.
A diagnostic gene signature to detect active mutant ERs
The team has been studying ESR1 gene translocations, which refer to the ER gene swapping a part of its sequence with genetic information from another gene. ER gene translocations create chimeric ER proteins, meaning the protein contains only half of the ER protein and the other half comes from different protein.
Some of the ER chimeras are extreme versions of mutant ERs because the drug-binding region, which is the same region estrogen binds to, is completely replaced with a region derived from another protein, to which neither the drug nor estrogen can bind. These ER chimeras trigger cancer activity in the absence of hormone.

“Not all ER translocations are active – some drive metastasis and resistance to treatment, but others do not,” Gou said. “To be able to determine whether any particular ESR1 translocation can promote disease progression, we developed a diagnostic genetic signature that detects the presence of an active ESR1 chimeric protein.”
With support from the National Cancer Institute’s PDXnet program, the team used genomics and transcriptomics to annotate 20 mouse models of ER+ patient-derived tumors that demonstrated different degrees of dependence on estrogen for growth. In this data set, a 24-gene signature detected the presence of an active ESR1 fusion, but interestingly also common point mutations in ESR1. These findings were replicated in data from a human metastatic breast cancer cohort. The team therefore called their 24-gene signature the MOTERA score for “Mutant or Translocated Estrogen Receptor Alpha.”
This work is significant in the area of precision medicine, as it can provide specific details about a tumor that can help guide a more precise selection of personalized, tumor-specific treatments.

“In the future, a patient’s cancer cells could be analyzed and, once the MOTERA score indicates the presence of an ER mutation or translocation, then the tumor cells would be further studied to more precisely determine what kind of ER mutant or translocation is present. This would help guide the selection of a personalized, optimal treatment,” said co-author, Dr. Charles E. Foulds, assistant professor at Baylor’s Lester and Sue Smith Breast Center.
The findings are published in Cancer Research.
Ellis and Foulds are members of the Dan L Duncan Comprehensive Cancer Center at Baylor. Other contributors to this work include Meenakshi Anurag, Jonathan T. Lei, Beom-Jun Kim, Purba Singh, Sinem Seker, Diana Fandino, Airi Han, Saif Rehman, Jianhong Hu, Viktoriya Korchina, Harsha Doddapaneni, Lacey E. Dobrolecki, Nicholas Mitsiades, Michael T. Lewis, Alana L. Welm, Shunqiang Li and Adrian V. Lee. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, Yonsei University Wonju College of Medicine, University of Cambridge, University of Utah, Washington University School of Medicine in St. Louis, University of Pittsburgh and University of Michigan.
See the publication for a complete list of the sources of support for this work.