Llama power: can tiny llama nanobodies improve norovirus anti-viral therapies?
Human noroviruses cause acute gastroenteritis, a global health problem for which there are no vaccines or antiviral drugs. Although most healthy patients recover completely from the infection, norovirus can be life-threatening in infants, the elderly and people with underlying diseases. Estimates indicate that human noroviruses cause approximately 684 million illnesses and 212,000 deaths annually.
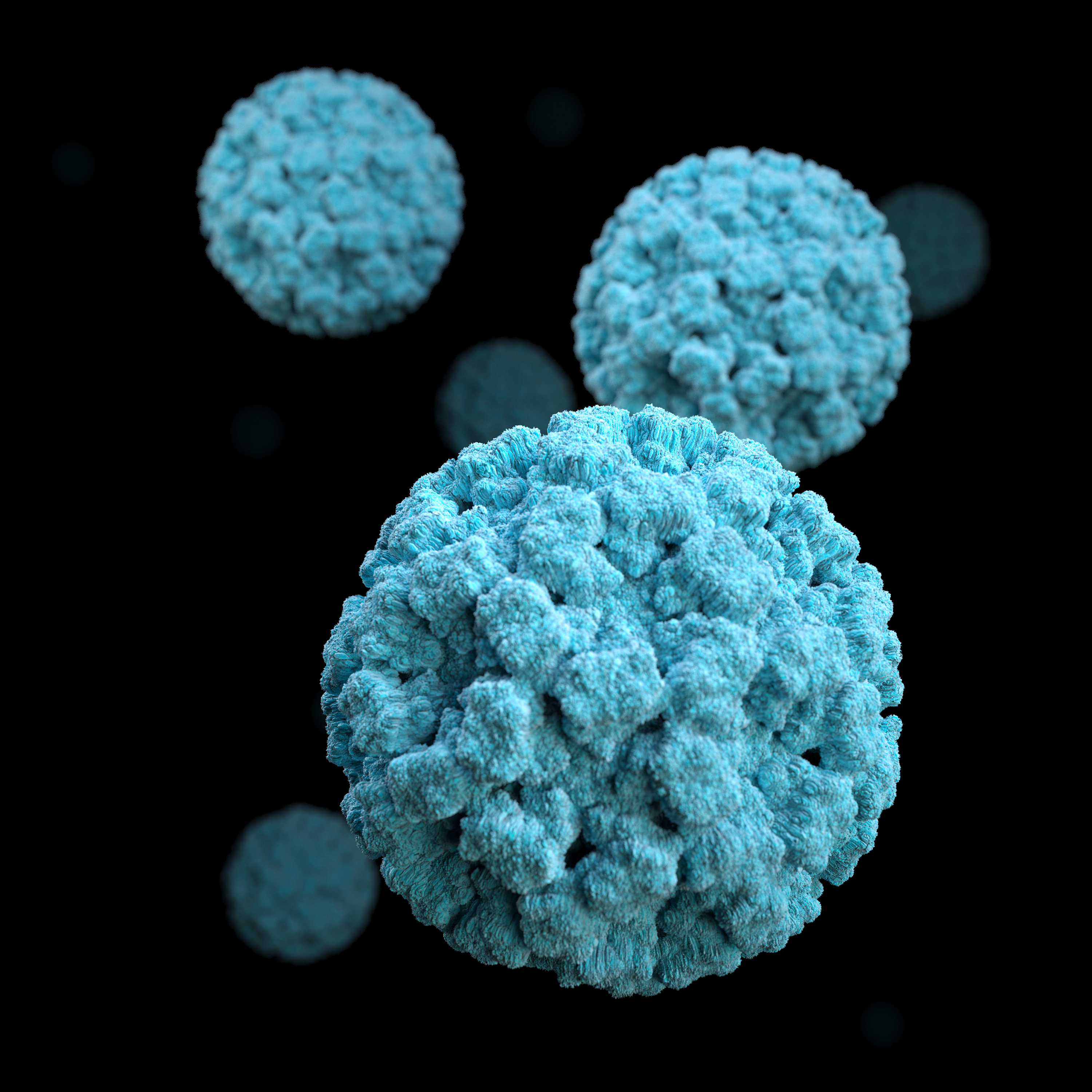
“Human noroviruses are highly diverse,” said first author Dr. Wilhelm Salmen, a graduate student in Dr. B V Venkataram Prasad’s lab while he was working on this project and currently a postdoctoral fellow at the University of Michigan. “Noroviruses are categorized into 10 groups, of which groups GI, GII, GIV, GVIII and GIX infect humans. Viruses in the GII.4 subgroup are the most predominant in human populations.”

Noroviruses also are notorious for periodically giving rise to new variants, particularly those of GII.4 norovirus, that can evade the immune response the body has developed against previous variants, like some flu viruses and coronaviruses do. The diversity of norovirus groups and the recurring emergence of new variants are some of the factors challenging the development of effective preventive and therapeutic approaches to control this serious disease.
In the current study published in the journal Nature Communications, Salmen, Prasad and their colleagues investigated a novel strategy to neutralize human noroviruses. They tested whether tiny antibodies produced by llamas, called nanobodies, could effectively neutralize human norovirus infection in the lab. The unexpected findings reveal that nanobodies could be developed as a therapeutic agent against human norovirus.
Llama nanobodies may give an upper hand

Llamas and related animals such as camels and alpacas, produce antibodies for protection against disease just like people do. However, compared to people’s antibodies, llama’s are about a tenth of the size of human antibodies. Llama’s nanobodies have been developed against viruses such as those causing hepatitis B, influenza, human immunodeficiency, polio and other diseases.
“Our collaborators from Argentina, Dr. Marina Bok and Dr. Viviana Parreño at the Institute of Virology and Technology Innovation, had prepared nanobodies from llamas that were inoculated with human norovirus-like particles from different strains,” Salmen said. “We worked with one nanobody named M4, which bound to the predominant GII.4 strain, testing its ability to neutralize different norovirus strains, that is, to prevent them from infecting human cells.”
The researchers tested the ability of the nanobodies to prevent live viruses from infecting human intestinal organoids or mini guts grown in the lab. Mini guts are models of human intestinal cells, closely representing actual small intestine tissue and its functions, that enable scientists to study how noroviruses work and to test potential therapies.

“It was really unexpected to see that the M4 nanobody not only interacted and neutralized the currently circulating pandemic GII.4 strain but also its older variants,” said Prasad, Alvin Romansky Chair in Biochemistry and professor in the Verna and Marrs McLean Department of Biochemistry and Molecular Pharmacology and the Department of Molecular Virology and Microbiology at Baylor. He also is a member of Baylor’s Dan L Duncan Comprehensive Cancer Center and corresponding author of the work.
The researchers used crystallography and other techniques to look closely at the interactions between nanobodies and noroviruses to try to understand how the M4 nanobody recognizes and neutralizes a variety of noroviruses when they expected it would recognize only the GII.4 strain used to generate M4.
We discovered that this little nanobody can recognize a part of the norovirus that all the different noroviruses that we tested have in common,” Salmen said.
Norovirus particles: a dynamic structure
The team discovered that the M4 nanobody recognized a hidden pocket in the norovirus particles that would be exposed only when the particles underwent a structural change. “The traditional thinking is that viral particles are in a very stable compact state, but in reality these particles ‘breathe’ considerably,” Salmen said. “Recent studies have shown that the structure of norovirus particles is dynamic, alternating between a resting or compact conformation and a raised conformation.”
“We think that the raised state is important for the virus to bind to cells and infect them,” Prasad said. “We also think that when the viral particles are in the raised state, the hidden pocket is exposed and available for the nanobody to bind to it and, acting like a wedge, to keep the particle in an elevated, potentially unstable state, preventing it from collapsing back down into the compact, more stable resting state.”
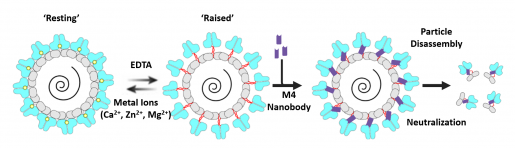
“Our findings suggest that trapping the viral particles in an elevated, unstable state disassembles the particles, which kills the virus. This effectively would stop the infection as it blocks the transmission chain, preventing the virus from spreading from cell to cell,” Salmen said.

“This study is also remarkable in confirming that the human norovirus must change its 3D conformation, from compact to raised, to infect people,” said co-author Dr. Mary Estes, Distinguished Service Professor of Virology and Microbiology and Cullen Foundation Endowed Chair at Baylor. She also is a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “Also, this work reveals the importance of considering viral particle dynamics when designing vaccines.”
Other contributors to this work include Liya Hu, Natthawan Chaimongkol, Khalil Ettayebi, Stanislav V. Sosnovtsev, Kaundal Soni, B. Vijayalakshmi Ayyar, Sreejesh Shanker, Frederick H. Neill, Banumathi Sankaran, Robert L. Atmar and Kim Y. Green. The authors are affiliated with one of the following institutions: Baylor College of Medicine, Institute for Virology and Technology Innovation-Argentina, National Institutes of Health and Lawrence Berkeley Laboratory.
This study was supported in part by the National Institutes of Health grants P30 GM124169-01, P30 CA125123 and P01 AI057788, and a grant from the Robert Welch Foundation (Q1279). Further support was provided by a Fulbright Program grant and by the Division of Intramural Research, NIAID, NIH.
Follow From the Labs on Twitter @BCMFromtheLabs.



