A deeper understanding of urinary tract infections reveals opportunity to fight them back
Urinary tract infections (UTI) are not only the most common type of infection, but are typically recurrent and tend to give rise to antibiotic-resistant bacteria, a serious medical concern.
Looking to identify novel strategies to combat this condition, the lab of Dr. Indira Mysorekar, E. L. Wagner Endowed Professor of Medicine-infectious diseases at Baylor College of Medicine, previously at Washington University School of Medicine, has taken a closer look at the cellular and molecular events following UTI and uncovered details that may lead to improved ways to manage this condition.
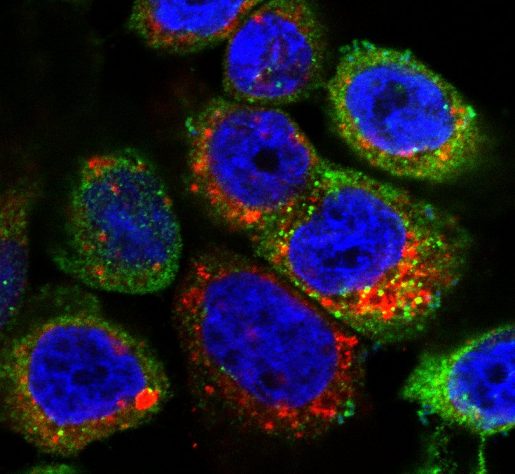

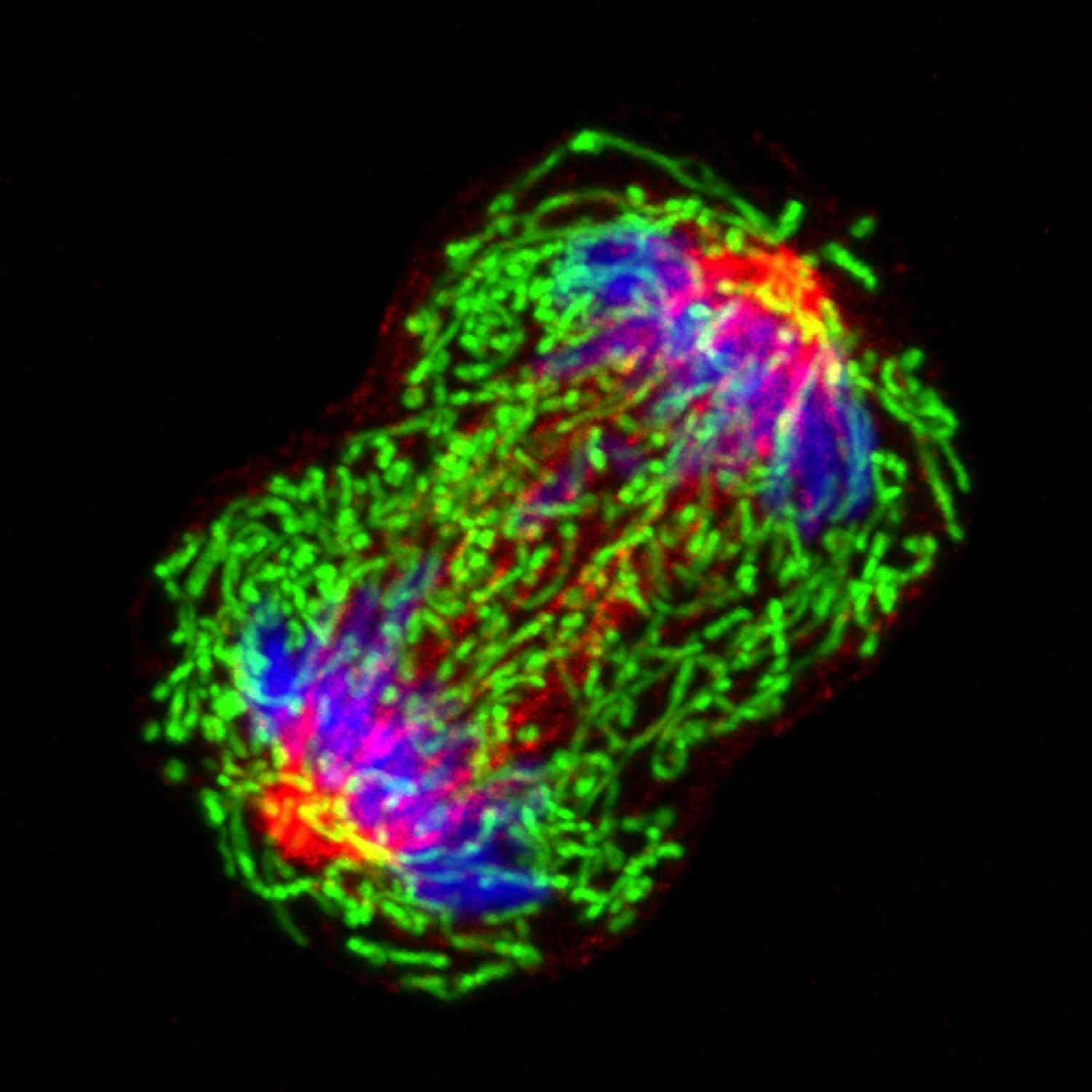
“More than 85% of UTI are caused by uropathogenic E. coli (UPEC), bacteria that can attach to the surface of the epithelial cells lining the inside of the bladder, called urothelial cells,” Mysorekar said. “Attached UPEC can then enter the urothelial cells, where they reproduce. In the current study, we looked at how urothelial cells fight back UPEC invasion and proliferation while preserving their integrity, which is essential for proper bladder function.”
A dynamic balance of responses
Working with urothelial cells grown in the lab, the team discovered that a precise sequence of events followed UPEC invasion of urothelial cells. First, in the early hours after their infection, urothelial cells defended themselves by producing reactive oxygen species (ROS), highly active compounds that kill bacteria. However, if sustained, ROS also can damage urothelial cells, which would be detrimental for the bladder.
“We found that accumulation of ROS activated an anti-ROS response in urothelial cells, called the NRF2 pathway, that minimized the damage excess ROS could cause to the urothelial cells,” said first author of the work, Dr. Chetanchandra S. Joshi, a postdoctoral associate in the Mysorekar lab.
The NRF2 protein is located in the cytoplasm of the cells bound to another protein called KEAP1.
“When ROS reaches a certain level, NRF2 separates from KEAP1 and goes into the nucleus of the cell, where it activates a series of genes. Some of these genes produce proteins that block ROS, and some produce proteins that limit inflammation,” Joshi said. “Interestingly, one of the genes NRF2 activates is Rab27b, which promotes the elimination of UPEC from urothelial cells.”
Together, these coordinated events mediate the elimination of UPEC while preserving the integrity of the cells attacked by the bacteria.”
A potential new way to combat UPEC
Understanding the process that follows a UPEC infection revealed a potential new strategy to combat the condition. “We learned that active NRF2 was involved in both neutralizing ROS, which helped protect urothelial cells, and eliminating UPEC,” Joshi said. “These findings suggested that a drug that activated NRF2, such as DMF, might help clear UPEC infections.”
DMF is FDA-approved to treat inflammatory conditions, such as multiple sclerosis, by dampening the inflammatory response.
“Working with an animal model of UTI, we showed that treatment with DMF activated NRF2, dampened the immune response, limited the level of damage the bacteria caused to urothelial cells and promoted activation of RAB27B, which removed bacteria from the bladder,” said Mysorekar, who also is professor of molecular virology and microbiology at Baylor. “Our findings support further exploration of this approach as a potential treatment for UTI.”
Women tend to have recurrent UTI, which can lead to chronic inflammation, extensive bladder mucosal damage and chronic infection. Continued antibiotic treatment also negatively affects the microbiome, the ‘good bacteria’ of the body, and promotes the development of antibiotic-resistant bacteria.
The most exciting part about this work was identifying a non-antibiotic-based therapy that contained the infection and reduced inflammation,” Mysorekar said.
“Although much work is needed before it reaches the clinic, treatment with DMF has the potential of helping millions of women affected by this condition.”
For all the details about this work, refer to the publication in Cell Reports.
Other contributors to this work include Amy Mora and Paul A. Felder, formerly at Washington University School of Medicine.
This work was supported in part by the National Institutes of Health grants R01AG052494, R01DK100644 and P20 DK119840.