Loss of gene Prss50 results in abnormal sperm tail development and infertility
One in ten men of reproductive age are infertile. Most infertile men have semen abnormalities, including impaired sperm motility in 51% of cases and/or impaired morphology or altered sperm shape in 54% of patients. Abnormalities in the sperm head have received more attention than those of the tail or flagella.
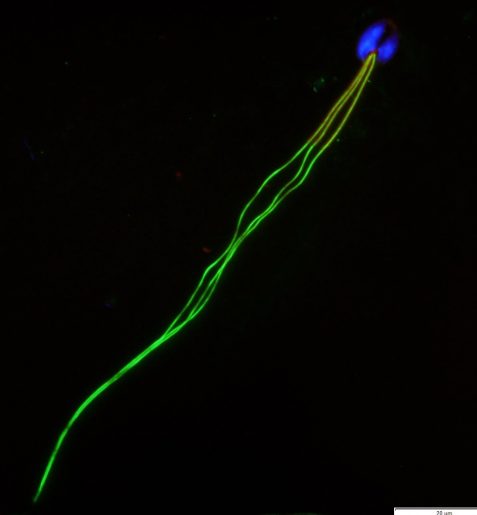
“The sperm tail is important for fertility because it propels the sperm in its journey to fertilize the egg,” said Dr. Carolina Jorgez, assistant professor of urology at Baylor College of Medicine. “My lab investigates male infertility in animal models and human samples. In the current study, we focused on sperm tail abnormalities.”

Some infertile men present with multiple morphological anomalies of the flagella (MMAF). The defects include absent, short, bent, coiled or irregular tails, which seriously impair motility. More than 20 genes have been associated with MMAF syndrome, suggesting the involvement of complex molecular mechanisms and challenging scientists looking to identify the cause.
“Understanding what causes infertility in men is not only important for the patient but also for his progeny because some genetic defects can be transmitted to the offspring,” said Jorgez, a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “Importantly, identifying genetic causes of infertility can lead to the development of screening tools to facilitate the specific diagnosis of the condition, helping patients make informed decisions regarding reproduction.”
In the current study, Jorgez and her colleagues investigated the contribution of an enzyme called protease Prss50, which is enriched both in human and mouse testis. Other proteases in the same family of Prss50, including Prss37 and Prss55, are essential for male fertility, but the role of Prss50 in fertility has not been studied before.
Jorgez and her team became interested in Prss50 after they discovered that 5% of the infertile men they studied had lost both copies of the gene. On the other hand, men having just one copy of the gene had no infertility problems.
Lack of Prss50 genes severely affects fertility in a mouse model
To investigate the contribution of Prss50 to male fertility, the researchers first developed a mouse model in which both copies of the gene were absent. Then, they assessed the fertility of these Prss50 knockout mice by comparing it to that of controls, animals carrying both copies of the gene. They found that mice lacking both copies of Prss50 were severely subfertile, producing 67% fewer pups than control mice. A closer look at Prss50 knockout mice revealed defects at multiple levels.
“When we looked at sperm production, we were a little disappointed at first because the mice lacking both genes had as much sperm as the controls,” Jorgez said. “But when we looked closely at the sperm under the microscope, we realized that about 70% of the sperm had abnormal morphology, compared to 28% in control mice.”
Defective sperm included having bent tails, heads with no tails and multiple tails, in some cases with multiple heads. They did not observe abnormalities in head morphology.
The researchers also found a sperm abnormality that had not been described before, a conjoined sperm, which had two heads, each on an opposite end of the tail.
“Conjoined sperm can move initially, but each head is pulling its own way, and usually after 15-20 min they stop moving,” Jorgez explained. “This abnormal sperm morphology has not been reported in humans, but we anticipate that it can exist. It was the most severe abnormal sperm morphology we observed. It was rare, we found it in 1% of the sperm.”
Lack of gene Prss50 results in untimely gene activation
Jorgez and her team investigated the molecular mechanism mediating abnormal sperm tail morphology in Prss50 knockout mice. They discovered that lacking Prss50 disrupts the timely activation of certain genes, and this leads to abnormal sperm development.
“Some genes have to be turned off at the beginning of sperm development and turned on at the later stages for normal sperm to form,” Jorgez explained. “We found that, in the absence of Prss50, genes that should be turned off at the beginning are turned on instead. We propose that the untimely activation of those genes is disrupting normal sperm development. We found that protease Prss50 is involved in regulating the timing of the expression of these genes, and its absence disrupts the process.”
Jorgez explained that defects in mechanisms that regulate gene expression, such as the one described here, can potentially affect the offspring.
“It’s important for men to know what specific problem is causing the infertility,” Jorgez said. “Current male fertility diagnostic tools are limited and not widely available.”
Our findings are a step toward identifying a panel of genes to screen for male infertility. I envision one day having a test looking at these genes, helping parents make informed decisions about having children.”
Learn all the details of this work in the journal Development.
Other contributors to this work include Jason M. Scovell, Juan C. Bournat, Adam T. Szafran, Minerva Solis, Joshua Moore, Armando Rivera, Ching H. Chen, Jason Zhang, Nathan Wilken and Abhishek Seth. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, Texas Children’s Hospital and Cleveland Clinic.
This research is supported by the National Institutes of Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development (grant 1R01HD100985) and the National Institute of General Medical Sciences (grant T32GM088129). Further support was provided by the Multidisciplinary K12 Urologic Research Career Development Program (K12DK0083014).