Platelets mediate a beneficial allergic response that protects against yeast asthma
We think of allergic diseases as unnecessary aberrant immune responses that do more harm than good. However, new evidence by an international team led by researchers at Baylor College of Medicine shows that some allergic reactions provide necessary protection against highly invasive and potentially lethal infectious agents, such as yeast.
“A common yeast called Candida albicans is a well-known cause of human allergic airway diseases, such as asthma,” said corresponding author Dr. David B. Corry, professor of medicine-immunology, allergy and rheumatology and Fulbright Endowed Chair in Pathology at Baylor. “However, the mechanisms by which yeast triggers asthmatic reactions are poorly understood.”

In addition to yeast, mold also is an important cause of human allergies. Corry and his colleagues had previously shown that mold triggers an allergic immune response by producing a type of enzymes called proteases. The enzymes activate T helper type 2 (Th2) and T helper type 17 (Th17) immune cells, which mediate the allergic response.
Mold and yeast are two kinds of fungi. “Candida also makes proteases, so we hypothesized that its proteases, like those of mold, also would be involved in promoting an allergic immune response,” said Corry, a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “We actually found something completely different.”
A new role for candidalysin

“Working with a mouse model of yeast-triggered asthma, we discovered that Candida proteases do not drive Th2 and Th17-mediated allergic immune responses. Instead, Candida promotes these types of immune responses by producing the peptide candidalysin,” said co-first author Dr. Yifan Wu, postdoctoral associate in the Corry lab.
Peptides, like proteins, are made of amino acids linked together like beads on a necklace, but include fewer amino acids than proteins. Candidalysin was originally described as a peptide toxin unique to Candida, capable of lysing or disintegrating cells. In this study, Corry, Wu and their colleagues found that candidalysin also can trigger an allergic immune response and the mechanism that mediates it.
The researchers discovered that candidalysin binds to the receptor GP1b-alpha, a molecule on the surface of platelets. This binding prompted the platelets to release a protein called Dkk-1, which in turn drove Th2 and Th17 allergic responses that correlate with reduced lung yeast burdens in experimental animal models.
Our results show that when the animal models produce an asthmatic response, they clear away Candida in the lungs very efficiently,” Wu said. “On the other hand, in the absence of platelets, there is no allergy response and Candida spreads unchecked, leading to a quick death.”
Supporting the role of platelets in the immune response
Platelets are well-known for their role mediating blood clotting to stop or prevent bleeding. In this study, Corry, Wu and their colleagues are the first to provide molecular evidence for another role of platelets as active contributors to the immune response.
When Candida enters the airway, the authors explain, platelets respond in two ways to combat the infection. On one side there is a Candida infection that very rapidly becomes invasive as the yeast burrows through lung tissue, rupturing and entering blood vessels from where it can migrate to other places in the body. In response to this invasion, the platelets activate their blood clotting function to reduce Candida’s advances.
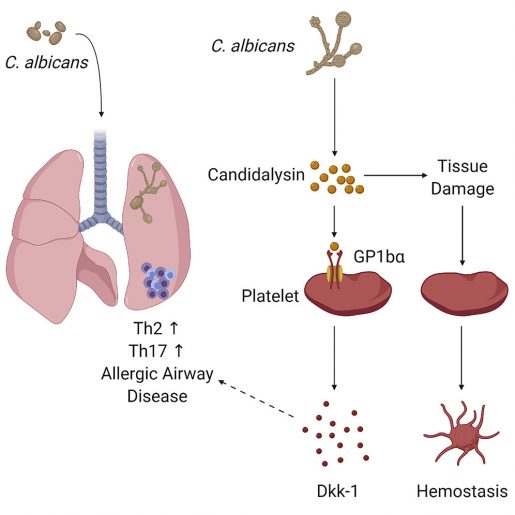
“Independently from their clotting function, we found that platelets respond to candidalysin by promoting the development Th2 and Th17-mediated allergic immune responses that confer immunological protection against Candida’s invasion,” Wu said. “This molecular pathway supports the role of platelets in the immune response.”
“We are most excited about crystalizing the notion that platelets are essential immune cells and the possibility of their roles in other conditions,” Corry said.
Read the complete report in the journal Immunity.
Other contributors to this work include co-first author Zhimin Zeng, Yubiao Guo, Lizhen Song, Jill E. Weatherhead, Xinyan Huang, Yuying Zeng, Lynn Bimler, Cheng-yen Chang, John M. Knight, Christian Valladolid, Hua Sun, Miguel A. Cruz, Bernhard Hube, Julian R. Naglik, Amber U. Luong and Farrah Kheradmand. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine; The First Affiliated Hospital of Sun Yat-sen University, China; Michael E. DeBakey VA Center for Translational Research on Inflammatory Diseases, Houston; Hans Knoell Institute Jena, Germany; Friedrich Schiller University, Germany and University of Texas Health Science Center, Houston.
Support for this study was provided by the US National Institutes of Health grants T32AI053831, R01HL117181, HL140398, R01AI135803 and R41AI124997; VA Office of Research and Development grant I01BX004828; the National Natural Science Foundation of China grant 81770024 and the Project of Department of Finance of Guangdong Province grant 20160907. This project was further supported by the Cytometry and Cell Sorting Core at Baylor College of Medicine with funding from the CPRIT Core Facility Support Award (CPRIT-RP180672), the NIH (CA125123 and RR024574) and by grants from the Wellcome Trust (214229_Z_18_Z), National Institutes of Health (R37-DE022550), the NIH Research at Guys and St. Thomas’s NHS Foundation Trust and the King’s College London Biomedical Research Centre (IS-BRC-1215-20006). Additional support was provided by the Deutsche Forschungsgemeinschaft (German Research Foundation) under Germany’s Excellence
Strategy (Balance of the Microverse Cluster – EXC 2051 – Project-ID 390713860) and the Collaborative Research Centre CRC/TR 124 FungiNet project C1.