Atf3 and Rab7b genes drive regeneration in mature cells
When an injury occurs, damaged cells need to be replaced. Stem cells, known as the go-to cells when new specialized cells need to be produced, are rare in adult tissues, so the job often falls to differentiated, or mature, cells.

Dr. Jason Mills and his lab have been working on identifying the genes driving mature cells to return to a regenerative state, a process called paligenosis.
“My lab has been promoting the idea that given that cells in all organs use similar functions like mitosis and apoptosis, there’s likely to be a conserved genetic program for how mature cells become regenerative cells,” said Mills, senior author of the study and professor of medicine – gastroenterology, pathology and immunology and molecular and cellular biology at Baylor. The research was conducted while his lab was at Washington University School of Medicine in St. Louis.
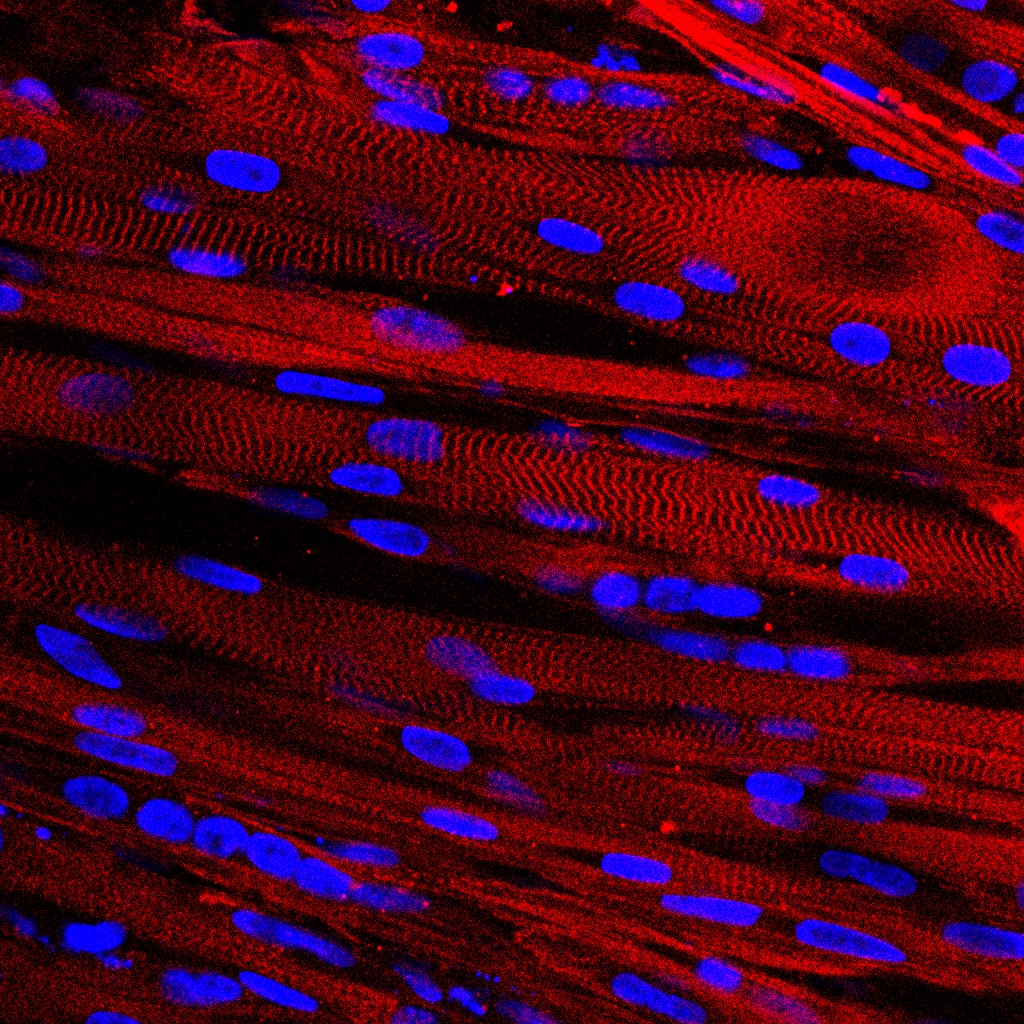
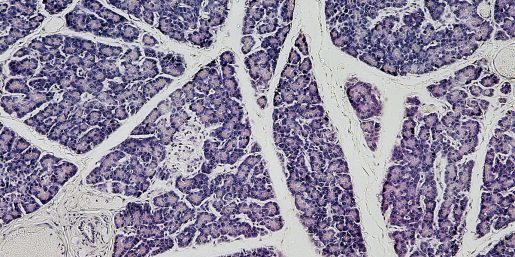
To begin paligenosis and reenter the cell cycle, mature cells must first go through the process of autodegredation, breaking down larger structures used in specialized cell function. Mills and his team, led by first author Dr. Megan Radyk, a postdoctoral associate at the Washington University School of Medicine in St. Louis at the time of research, found that the genes Atf3 and Rab7b are upregulated in gastric and pancreatic digestive-enzyme-secreting cells of mice during autodegredation, and return to normal expression before mitosis.

The researchers showed that Atf3 activates Rab7b, which directs lysosomes to begin dismantling cell parts not needed for regeneration. But when Atf3 was not present, Rab7b did not trigger autodegredation.
Repetitive injury, paligenosis and cancer development
The team also found Atf3 and Rab7b expression were consistent in paligenosis across other organs and organisms. Similar gene expression also appeared in precancerous gastric lesions in humans. According to Mills, the discoveries in this research are foundational to understanding how repetitive injury and paligenosis may impact cancer.
“The more tissue damage you have, the more you’re calling mature cells back into regeneration duty,” said Mills, co-director of the Texas Medical Center Digestive Disease Center. “There’s emerging evidence that, when these cells go through paligenosis, they don’t check for DNA damage well. The cells are storing DNA mutations when they return to their differentiated function. Over time, they become so damaged that they can’t go back to normal function and instead keep replicating.”
It’s our belief that paligenosis is at the heart of cancer development.”
This research also provides groundwork for potential therapeutic targets. Existing drugs like hydroxychloroquine can be used to inhibit autodegredation, therefore stopping paligenosis.
According to Mills, further study is required to determine whether drugs targeting autodegredation can be used in conjunction with cancer treatments to stop cells from replicating.
The complete study is published in EMBO Reports.
For a full list of authors, their contributions to this work and sources of support, see the publication.
By Molly Chiu