Of fruit flies, genes and aortic aneurysms
One of the goals of Dr. Hugo Bellen’s lab is to conduct genetic screens in the fruit fly to identify genes that cause neurodevelopmental and neurodegenerative disorders in people.

“We have identified 165 genes, mapped them and cloned them. The gene we studied here, ari-1, is one of these genes,” said Bellen, professor of molecular and human genetics and neuroscience at Baylor College of Medicine and an investigator at the Howard Hughes Medical Institute.
“We studied ari-1 because we wanted to investigate a possible connection with the gene parkin, linked to Parkinson disease,” said Dr. Nele Haelterman, a former graduate student in the Bellen lab. “The ari-1 and parkin genes are related, and the corresponding proteins they produce are enzymes that chemically modify many other proteins in ways that alter their functions.”

Working with fruit flies carrying mutations of ari-1, the researchers screened the flies’ tissues looking for anomalies.
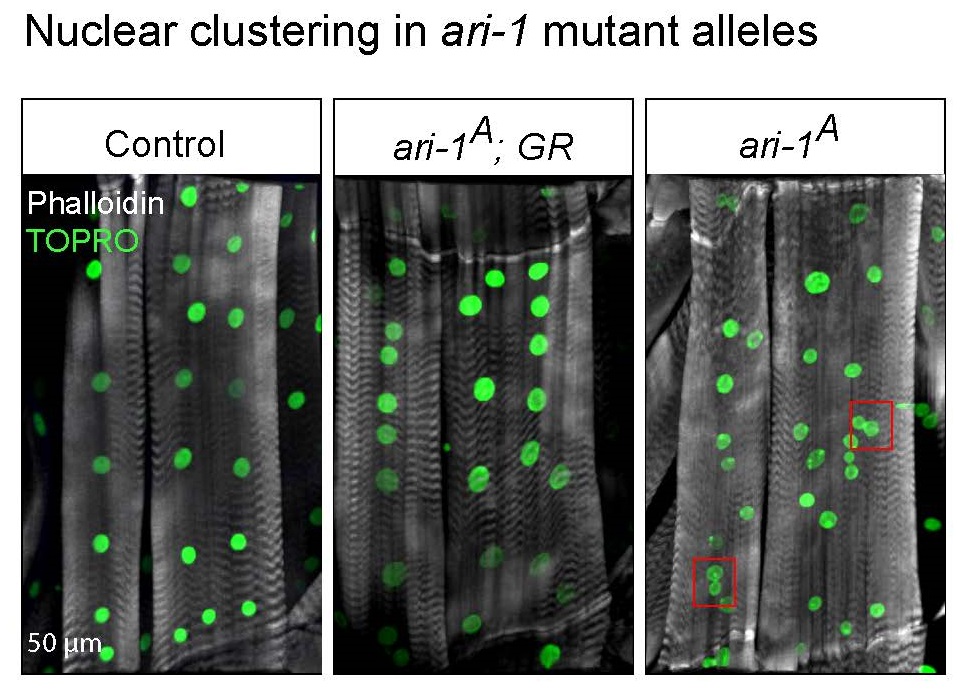
“We found that muscle cells from ari-1 mutants looked different from normal cells,” said Kai Li Tan, a graduate student in the Program in Developmental Biology in the Bellen lab. “Normal muscle cells have many nuclei that are evenly distributed in the cell. On the other hand, ari-1 mutant muscle cells display a clustering of nuclei similar to what one would observe in an injured or sick muscle. The nuclei also had abnormal shapes.”

Normal fly muscle cells maintain their nuclei evenly distributed using a network of complex protein ‘bridges,’ called cytoskeleton. The cytoskeleton is connected to a similar array inside each nucleus (the nucleoskeleton) by the LINC protein complex. This array of protein connections not only keeps nuclei in their proper location, but it also allows the inside of the cell, including the nuclei, to sense changes, such as mechanical tension changes that occur in their external environment.
“Mutants of these different proteins have been known to cause nuclear clustering,” Tan said. “We tested whether the LINC complex was affected in ari-1 mutants and found that the mutants do modify the protein Koi in the LINC complex in a manner that disrupts the mechanism that keeps the nuclei in place.”
The researchers also found an interesting connection between the proteins Ari-1 and Parkin.
“We found that the proteins can form complexes with themselves and with each other and that the functions of Ari-1 and Parkin overlap to some degree,” said Bellen, who also is a member of the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital. “If Ari-1 is missing, Parkin can take over its function, but not as well as Ari-1; the fruit flies present a mild defect. It works the other way, too. When Parkin is missing, Ari-1 can compensate for its absence to some degree.”
The fruit fly-human connection
The researchers looked to connect the fly gene ari-1 to a human condition via GeneMatcher, a web-tool developed as part of the Baylor-Hopkins Center for Mendelian Genomics for rare disease researchers. Similar to online dating websites that match couples, GeneMatcher allows researchers to find others that are interested in the same genes they are working on.
GeneMatcher connected the Bellen group with the group of Dr. Dianna Milewicz, division director, professor and vice chair of medical genetics at the University of Texas Health Science Center at Houston McGovern Medical School. She also is a co-author in this study.

Milewicz is interested in finding new genes linked to aortic aneurysms. Her group has screened hundreds of families with early onset aortic aneurysms to try to find new genes that could help assess early the risk of the disease. Aortic aneurysms are potentially life threatening conditions in which the walls of the body’s main artery, the aorta, can weaken, bulge and potentially rupture, a fatal condition.
The Bellen and Milewicz labs began a collaboration to explore a connection between human ari-1 (ARIH1) and aortic aneurysms. They identified rare variants of ARIH1 in patients. The variants are associated with cerebrovascular and aortic aneurysms and confirmed that patient aortic tissue samples have abnormally shaped nuclei in aortic smooth muscles. Further, laboratory experiments reducing ARIH1 expression in normal human smooth cells resulted in nuclei changing their shape to that observed in the tissues taken from patients.
Given that proper sensing of mechanical tensions affecting the aorta is important to prevent aneurysms, the researchers argue that the defects in ARIH1 that lead to disruption of mechanical sensing mechanisms implicate ARIH1 in aortic and cerebrovascular disease.
Read all the details about this study in the journal Developmental Cell.
Other contributors include Callie S. Kwartler, Ellen S. Regalado and Dong-Chuan Guo with the University of Texas Health Science Center at Houston McGovern Medical School, Pei-Tseng Lee, Sonal Nagarkar-Jaiswal, Lita Duraine, Michael F. Wangler and Guang Lin with Baylor College of Medicine, Michael Bamshad and Deborah A. Nickerson with the University of Washington.
This work was supported in part by NIH grants R24OD022005 and R01GM067858, the National Heart, Lung and Blood Institute (R01HL109942 and P01HL110869), the John Ritter Foundation, and the Henrietta B. and Frederick H. Bugher Foundation. Sequencing was provided by the University of Washington Center for Mendelian Genomics (UW-CMG) and was funded by the National Human Genome Research Institute and the National Heart, Lung and Blood Institute grantUM1HG006493. Microscopy at Baylor College of Medicine is supported by IDDRC (U54 HD083092) from the Eunice Kennedy Shriver National Institute of Child Health and Human Development.