How understanding tumor diversity can potentially improve the outcomes of patients with lung cancer
[See video below]
“People may think of lung cancer as one disease, but lung cancer is a collection of diverse subtypes of cells and each subtype may respond differently to the same therapy,” said Dr. Chad Creighton, associate professor of medicine and member of the Dan L Duncan Comprehensive Cancer Center Division of Biostatistics at Baylor College of Medicine.

The heterogeneity of cancer poses major challenges to physicians and scientists and suggests that personalized therapies can potentially improve the outcomes of patients with lung cancer. However, how to best design such an approach is not always clear.
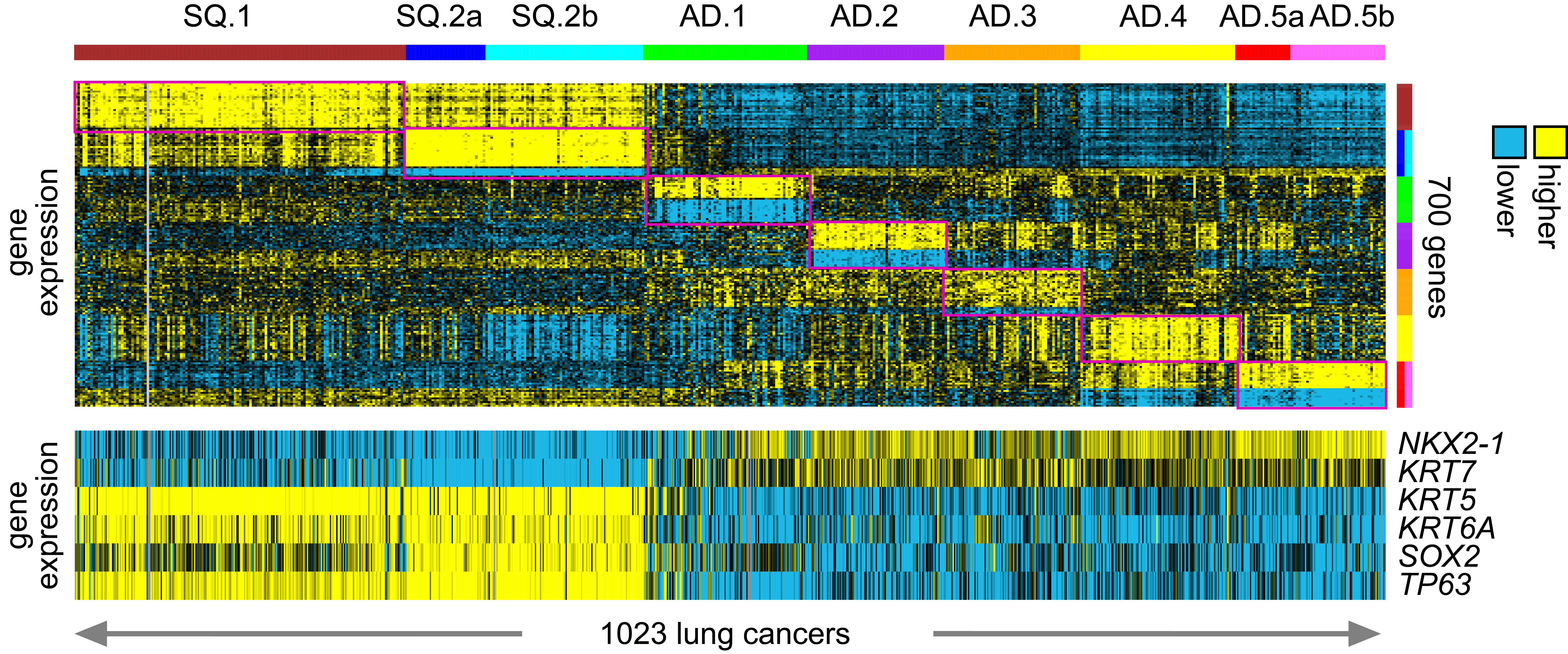
One approach was carried out by a team of scientists from Baylor College of Medicine and the University of Texas MD Anderson Cancer Center. They analyzed vast amounts of molecular data from a set of more than 1,000 non-small cell lung cancers that allowed them to break down the cancers into distinct subtypes, each with its own molecular profile and potentially different response to therapy.
By using molecular data – which would inform scientists, for instance, about which genes and proteins the cancer expresses and changes in its DNA– the researchers were able to tease out the different subtypes and have a more refined system of classification for non-small cell lung cancer.
“Any two given lung cancers may have very different molecular profiles,” said Creighton. “One would have certain genes turned on and produce certain proteins while the other cancer would have different genes turned on and express different proteins. This would imply that one cancer subtype might be more vulnerable to specific therapies while the other might be more susceptible to other therapies.”
Unraveling the complexity of the tumor microenvironment
The researchers found that the tumors are complex in more than one way. They are complex because of the diversity of alterations involving tumor cells, but also in the variety of non-tumor cells that are part of the tumor microenvironment.
“One big part of the study was to try to tease out the cells in the tumor that actually are not cancer,” said Creighton. “Those cells include lymphocytes – a type of immune cell that tries to attack the cancer. Lymphocytes showed up in the tumor profile and we found out that only specific subsets of lung cancer show this really strong pattern for lymphocyte infiltration. These cancer subtypes also show evidence for immune check point pathway, which is a pathway by which cancer cells learn to evade the immune system which normally would actively attack and kill cancer cells. Many cancers have learned some ‘tricks’ so they can turn on or off specific genes or proteins that allow them to evade the immune response.”
With these types of analyses the researchers identified specific subsets of cancer cells that they think might be more responsive to immunotherapy – a therapy to boost the body’s natural defenses to fight the cancer. Immunotherapy has had a lot of success in a subset of human cancers.
“The Cancer Genome Atlas, the cancer initiative that generated these data that we analyzed, involves data for over 10,000 cancers,” said Creighton. “That information is now in the public domain and we are currently going through all of the data, not just for lung cancer, but for all the cancer types. We call this pan cancer analysis. We are trying to find features that not just apply to one type of cancer, but across multiple types. Much of our work in the next year is going to be focusing on pan cancer work that might point at ways of treating multiple types of cancer.”
###
Other contributors to this work include Fengju Chen, Yiqun Zhang, Edwin Parra, Jaime Rodríguez, Carmen Behrens, Rehan Akbani, Yiling Lu, Jon Kurie, Don Gibbons, Gordon Mills and Ignacio Wistuba, who are affiliated with Baylor and/or the University of Texas MD Anderson Cancer Center.
Financial support for this project was provided by the National Institutes of Health (NIH) grant 2R01CA125123-09, MD Anderson’s Institutional Tissue Bank Award 2P30CA016672, The University of Texas Lung Specialized Programs of Research Excellence grant P50CA70907, Cancer Prevention and Research Institute of Texas (CPRIT) grant RP120713 C2, P2 and RP150405 and Department of Defense PROSPECT grant W81XWH-07-1-0306.
The results appear in Oncogene.