Deep brain stimulation induces more healthy neurons and sustained memory benefits in Rett animal models
A recent study from the lab of Dr. Jianrong Tang, associate professor of pediatrics – neurology at Baylor College of Medicine and principal investigator at the Jan and Dan Duncan Neurological Research Institute (Duncan NRI) at Texas Children’s Hospital shows that deep brain stimulation (DBS) of a specific brain region results in a significant and sustained improvement of memory in mouse models of Rett syndrome. Moreover, they found that this treatment promotes the generation of new healthy neurons in these mice.
The study, published in the journal Brain Stimulation, provides crucial preclinical evidence that supports the development of DBS as a feasible treatment for Rett syndrome patients.

Rett syndrome is a rare but devastating neurodevelopmental disorder that causes regression of motor, social, cognitive and language skills along with the development of autistic behaviors and seizures. It is caused by mutations in the X-linked gene that encodes the methyl-CpG-binding protein 2 (MeCP2) protein and thus, primarily affects young girls. Since the loss of cognitive skills is one of the tragic aspects of this condition, there has been intense interest in developing therapeutic approaches to mitigate this loss.
DBS induces significant and sustained improvements in learning and memory among Rett mice
DBS is a neurosurgical procedure that involves the placement of a medical device called a neurostimulator to send electrical signals through the electrodes that have been implanted in specific brain regions to treat various neurological conditions such as movement disorders (for example, Parkinson’s, tremors, dystonia), neuropsychiatric disorders (such as obsessive-compulsive disorders, depression), and epilepsy.
“Inspired by a study which showed that stimulation of the fornix region of the hippocampus improved memory in Alzheimer’s disease, a few years ago we performed DBS in this region in a mouse model of Rett syndrome and found that two weeks of a specific regimen of this treatment improved learning and memory in female Rett mice,” Tang said. “While those findings demonstrated the potential of DBS to mitigate learning and memory deficits in Rett mice, it was not clear how long the benefits of this treatment would last.”
Their goal in this study was to address that question.
Using well-established behavioral assays, the team discovered that DBS improved contextual fear memory in Rett mice, and these beneficial effects persisted for 6 to 9 weeks after the treatment. Further, repeated DBS – specifically in the fornix, not other brain regions – maintained the contextual memory improvement in Rett mice.
DBS induces the generation of new hippocampal neurons in adult Rett mice
Adult hippocampal neurogenesis – the process of forming new neurons in specialized regions of the hippocampus of adult animals – is critical for the neurons to “learn and remember.” Previous studies from the Tang lab revealed that right after the DBS treatment, many new neurons were born in specialized regions of the hippocampus. Since it takes between 2 to 5 weeks for newborn neurons to mature and functionally integrate into the neural networks, this correlated with improved contextual memory in these mice three weeks later.
The researchers found this close correspondence between the DBS-induced birth of new neurons and improvements in contextual memory persisted several weeks after the treatment as well.
DBS promotes a neuroprotective environment with healthy neurons
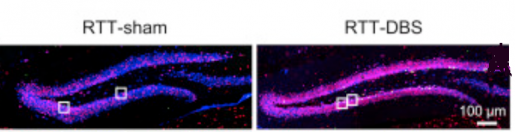
In female mice that carry one copy of defective MECP2, the expression of this gene is switched off/inactivated in some neurons. This means some newborn neurons in Rett mice will be healthy and have a non-defective MeCP2 while others may turn off/not express MeCP2. Interestingly, the team found that forniceal DBS preferentially induced the birth of more healthy neurons with normal MeCP2 gene in Rett mutants.
Moreover, DBS treatment also induced the production of brain-derived neurotrophic factor (BDNF), which is a neuroprotective molecule essential for the survival and growth of neurons as well as for their plasticity, that is their ability to learn and remember.
The findings from this study are very promising.
Our findings show that DBS not only results in a significant and long-lasting improvement to memory in Rett animal models, but it also promotes a favorable neuroprotective environment in the brain and leads to the production of more healthy neurons – which together suggests DBS could be an excellent therapy to treat Rett patients,” Tang said.
Qi Wang, Bin Tang, Shuang Hao, Zhenyu Wu and Tingting Yang were also involved in this study. They are affiliated with Baylor College of Medicine and the Duncan NRI.
The study was funded by the National Institute of Neurological Disorders and Stroke, a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to Baylor College of Medicine Intellectual and Developmental Disabilities Research Center, Neuroconnectivity Core, Circuit Modulation Core, Neurovisualization Core, In Situ Hybridization Core, the In Vivo Neurophysiology Core of the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital, the Chao Family Foundation and the Cockrell Family Foundation.
The original article by Rajalaxmi Natarajan, Ph.D. is posted here.
Follow From the Labs on Twitter @BCMFromtheLabs.