A new biological mechanism to regenerate and repair myelin
A study led by Dr. Hyun Kyoung Lee, associate professor of pediatrics – neurology at Baylor College of Medicine and investigator at the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital, reveals a new biological mechanism to regenerate and repair myelin, a protective sheath that insulates neuronal fibers and plays a vital role in ensuring rapid and accurate neurotransmission.
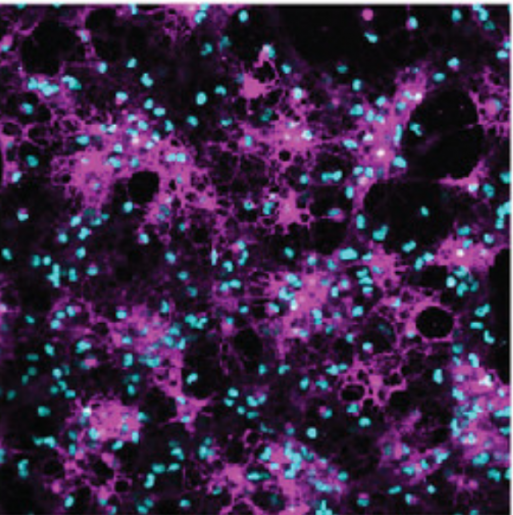
The team found novel roles for two proteins, dishevelled associated activator of morphogenesis 2 (Daam2) and CK2α kinase, in regulating myelin repair and regeneration. The study was published in the Proceedings of the National Academy of Science.
What we knew about myelin regeneration
Myelin is produced by a type of glial precursor cells called oligodendrocytes, which are among the most numerous cells in the nervous system. Damage or loss of myelin sheath is the hallmark of various neurological diseases in adults (such as multiple sclerosis) and infants (for example, cerebral palsy) and is common after brain injuries.

The Wingless (Wnt) signaling pathway is one of the key regulators of oligodendrocyte development and myelin regeneration. In certain diseased conditions and brain injuries, its levels are elevated in the white matter, which impairs myelin production by forcing oligodendrocytes to remain in a “stalled/quiescent state.”
A few years ago, Lee and others found that the glial protein Daam2, which is part of the Wnt signaling pathway, inhibits the differentiation of oligodendrocytes during development as well as myelin regeneration and repair. However, until now precise mechanisms underlying this process have remained a mystery.
A new mechanism to regenerate and repair myelin
To understand how Daam2 inhibits myelination, the team first needed to determine the regulation of Daam2 itself. Using biochemical approaches, they found that cells regulate Daam2 activity by tagging it with phosphate chemical groups, a process called phosphorylation, which affects which genes are turned off or on.
Intriguingly, we found Daam2 phosphorylation differentially affected distinct stages of oligodendrocyte development.”
In early stages, it accelerates the conversion of oligodendrocyte precursors to glial cells, but in later stages, it slows down their maturation and their ability to produce myelin,” said Lee, a member of Baylor’s Dan L Duncan Comprehensive Cancer Center.
Further analyses showed that protein CK2 is responsible for Daam2 phosphorylation. They confirmed that the subunit CK2α interacted with Daam2 in lab-cultured oligodendrocytes and phosphorylated it. Using laboratory cultured oligodendrocytes and mouse models, they found compelling evidence suggesting that CK2α promotes oligodendrocyte differentiation by phosphorylating Daam2.
A neonatal hypoxic-injury animal model revealed that CK2α mediated Daam2 phosphorylation, which plays a protective role in developmental and behavioral recovery after neonatal hypoxia, a form of brain injury seen in cerebral palsy and other conditions. Additionally, it facilitates remyelination after white matter injury in adult animals.
Together, these findings have identified a novel regulatory node connecting CK2α and Daam2 in the Wnt pathway that regulates stage-specific oligodendrocyte development and offers insights into a new biological mechanism to regenerate myelin.
“This study opens exciting therapeutic avenues we could develop in the future to repair and restore myelin, which has the potential to alleviate and treat several neurological issues that are currently untreatable,” Lee said.
The first author, Chih-Yen Wang is now an assistant professor in the National Cheng Kung University. Others involved in the study were Zhongyuan Zuo, Juyeon Jo, Kyoung In Kim, Christine Madamba, Qi Ye, Sung Yun Jung and Hugo J. Bellen. They are affiliated with Baylor College of Medicine and/or the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital.
This work was supported by grants from NIH/NINDS, the National Multiple Sclerosis Society, the Cynthia and Anthony G. Petrello Endowment, the Mark A. Wallace Endowment and the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health for the BCM IDDRC Neurobehavior and Neurovisualization Cores. GERM core at Baylor College of Medicine helped with mouse line generation. scRNA-sequencing was partially supported by the SCG core and GARP core.
The original article by Rajalaxmi Natarajan, Ph.D. is posted here.
Follow From the Labs on Twitter @BCMFromtheLabs.



researching in context with peripheral neuropathy