Prolonged infection and inflammation drain immune responses as we age
Like a savings account that can support our needs when the occasion calls, our body is born with cells called hematopoietic stem cells (HSCs) that collectively ensure lifelong production of blood and immune cells that protect us from infections.
Just as we can maintain or grow our savings account or use some of it to satisfy specific needs, HSCs can either reproduce to generate more stem cells or differentiate into distinct immune cell lineages to fulfill specific functions. Going in one direction or the other is an extremely critical decision that ensures that the body achieves the fine balance between having enough immune cells to fight invaders while still retaining enough HSCs to maintain future blood production.
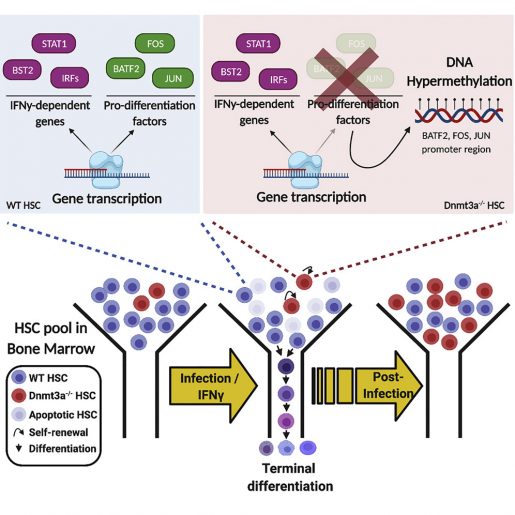
As we age, HSCs accumulate mutations that lead to genetically distinct subpopulations. This common phenomenon known as clonal hematopoiesis (CH) starts in early fifties and is frequently associated with loss-of-function mutations in the DNMT3A gene. CH is associated with a significantly higher risk of blood cancers, cardiovascular disease, stroke and all-cause mortality.
The laboratory of Dr. Katherine King, associate professor of pediatrics-infectious disease at Baylor College of Medicine and Texas Children’s Hospital has long been studying CH and in the current study they show for the first time that long-term infection and prolonged inflammation drive CH mediated by the loss of Dnmt3a function. In addition, the study offers key insights into the mechanism by which chronic inflammation leads to CH and demonstrates the critical role of DNMT3a in regulating normal HSC responses to infections.

How prolonged inflammation dampens the body’s immune response as we age
Previously, the King lab showed that chronic infection significantly impaired the ability of wild-type HSCs to remain in a quiescent stem cell state. Months-long exposure to a body-wide bacterial infection promoted extensive differentiation of HSCs.
“While this produced sufficient immune cells to fight infection, it also reduced the number of bone marrow HSCs by 90%,” said King, a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “In contrast, HSCs in mice lacking the Dnmt3a gene did not differentiate much. In fact, they underwent self-renewal to produce more HSCs.”
In the current study, the researchers tested their prediction that defective differentiation and increased duplication of Dnmt3a HSCs allowed them to overtake and outcompete normal HSCs when fighting chronic infections or facing long-term inflammatory conditions.
To test their hypothesis, researchers used a combination of experimental and mathematical modeling experiments to test how HSCs from Dnmt3a mutant mice respond to long-term infection and chronic inflammation. For experimental validation, they generated mosaic mice by transplanting a mixture of whole bone marrow from Dnmt3a-mutant mice and normal mice into irradiated mice.
In these mice, the team tracked how each subpopulation of HSC reduced its numbers or grew relative to one another over time when infected for several months with Mycobacterium avium bacteria.
Using this model that mimics chronic infection in humans, they found that long-term infection caused specific expansion of Dnmt3a-loss-of-function HSCs along with a concomitant reduction in their ability to differentiate into immune cells, which is contrary to the behavior exhibited by normal HSCs to chronic infection.
Moreover, compared to the normal HSCs, Dnmt3a HSCs were more resistant to exhaustion and were less sensitive to stress-induced apoptosis, also called ‘cell death,’ upon chronic infection. Collectively, this indicates how a minor population of Dnmt3a HSCs could eventually overtake a major population of normal HSCs in the presence of chronic infection.
Viral or bacterial infections and chronic inflammatory stress conditions, including tuberculosis, hepatitis, herpetic infections and inflammatory bowel disease, trigger the release of interferon gamma (IFNγ) by the immune system, which in turn, initiates a cascade of protective immune responses. The team found that, compared to wild-type HSCs, Dnmt3a-loss-of-function HSCs exhibited an entirely opposite set of cellular responses and global changes in gene expression patterns in response to IFNγ. These changes tended towards preserving or even increasing the numbers of stem cells at the expense of mounting an effective response against imminent invaders or stress.
“We are excited by the findings of this study which opens several areas of future investigations.”
We have shown for the first time how chronic inflammation due to long-term infections or autoimmune conditions, such as rheumatoid arthritis, ulcerative colitis or Crohn’s disease, dampen the body’s immune response as we age,” King said. “Moreover, our findings shed light on the critically important role of DNMT3a in modulating immune responses during chronic infection or stress and also can explain how aging and inflammation are linked to blood cancers.”
The study was published in the journal Cell Stem Cell.
Other authors involved in the study are Daniel Hormaechea-Agulla, Katie Matatall, Duy Le, Grant Challen, and Marek Kimmel. They are affiliated with one or more of the following institutions: Baylor College of Medicine, Rice University, Silesian University of Technology and Washington University School of Medicine.
Grants from National Institutes of Health, Dan L. Duncan Cancer Center, and Polish National Science Center supported this work.
Read more.