Six is better than two: assay assesses multiple cellular pathways at once
Scientists strive to have a better understanding of the complex biological processes involved in health and disease, and what they can learn usually goes hand-in-hand with the number, quality and type of measurements techniques provide.
“Cancer, for instance, usually originates through changes on many different genes and pathways, not just one, but currently most cell-based screening assays conduct single measurements,” said Dr. Koen Venken, assistant professor of biochemistry and molecular biology, and pharmacology and chemical biology at Baylor. “We thought that if we could see what happens to more than one cellular pathway at once, we could have a more complete picture of what goes on inside a cancer cell.”
To get a more detailed picture of the cellular processes that differentiate normal versus cancer cells, researchers resort to conduct several independent screening assays at the expense of time and additional cost.
In his lab at Baylor College of Medicine, Venken and his colleagues apply state-of-the-art synthetic biology, cell biology, genetics, genome engineering and transgenic technologies to have a better understanding of the processes involved in cancer.

“Our goal in this study was to measure multiple cellular pathways at once in a single biological sample, which would also minimize experimental errors resulting from conducting multiple separate assays using different samples,” said Venken, a McNair Scholar and member of the Dan L Duncan Comprehensive Cancer Center at Baylor.
Six is better than two
Dr. Alejandro Sarrion-Perdigones, first author of the paper, wanted to develop an experimental assay that would expand the number of molecular pathways that can be studied simultaneously in a cell sample.
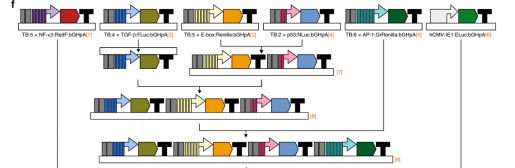
He focused on developing a multiplexed method – a method for simultaneously detecting many signals from complex systems, such as living cells. He developed a sensitive assay using luciferases, enzymes that produce bioluminescence. The assay includes six luciferases, each one emitting bioluminescence that can be distinguished from the others. Each luciferase was engineered to reveal the activity of a particular pathway by emitting bioluminescence.

“To engineer and deliver the luciferase system to cells, we used a ‘molecular Lego’ approach,” said co-author Dr. Lyra Chang, post-doctoral researchers at the Center for Drug Discovery at Baylor. “This consists of connecting the DNA fragments encoding all the biological and technological information necessary to express each luciferase gene together sequentially forming a single DNA chain called vector. This single vector enters the cells where each luciferase enzyme is produced separately.”
Treating the cells with a single multi-luciferase gene vector instead of using six individual vectors, decreased variability between biological replicates and provided an additional level of experimental control, Chang explained. This approach allowed for simultaneous readout of the activity of five different pathways (a control makes number six), compared to just one using traditional approaches, providing a much deeper understanding of cellular pathways of interest.
The new assay is sensitive, saves time and expense when compared to traditional approaches, reduces experimental error and can be adapted to any research field where the dual luciferase assay is already implemented, and beyond.
“In addition to applications in cancer research, as we have shown in this work, our multiplex luciferase assay can be used to study other cellular pathways or complex diseases across different research fields,” Venken said. “For instance, the assay can be adapted to study the effect of drugs on insulin sensitivity in different cell types, the immune response to viral infections or any other combinations of pathways.”
Interested in this new technology? Find all the details in the journal Nature Communications.
Other contributors to this work include Yezabel Gonzalez, Tatiana Gallego-Flores and Damian W. Young, all at Baylor.
This work was supported by start-up funds provided by Baylor College of Medicine, the Albert and Margaret Alkek Foundation and the McNair Medical Institute at The Robert and Janice McNair Foundation. Additional support was provided by March of Dimes Foundation grant #1-FY14-315, the Foundation For Angelman Syndrome Therapeutics grant FT2016-002, the Cancer Prevention and Research Institute of Texas grants R1313 and R1314 and the National Institutes of Health grants 1R21GM110190, 1R21OD022981 and R01GM109938.
The authors dedicate this work to the memory of Dr. Alejandro Sarrion-Perdigones, who passed away before the paper was published.