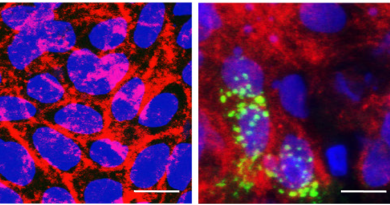
Mutant PPM1D gives stem cells a survival advantage
Scroll down to watch the video summary of this work.
Although chemotherapy can fight back cancer, it also has been associated with increased risk of leukemia years after the treatment. What leads to that association is not clear, but a recent report has provided some answers. The answers involve a gene called PPM1D, whose function in blood production was unknown. Furthermore, the implications of these findings can affect the choice of chemotherapies. The journey that led to these findings began on the bedside and then moved into the research lab, highlighting the importance of collaboration between clinical and basic science researchers.
The research team led by corresponding authors Dr. Margaret A. Goodell, director of the Stem Cells and Regenerative Medicine Center and professor of pediatrics at Baylor College of Medicine, and Dr. Koichi Takahashi, assistant professor in the department of leukemia at the University of Texas MD Anderson Cancer Center, initially found that PPM1D mutations turn up frequently in the blood of patients who get leukemia years after they first received chemotherapy for a previous disease. This finding sparked the question: how do PPM1D mutant cells become dominant when compared to their normal counterparts in the bone marrow? Do they ‘win’ because they are stronger and better at making more of themselves, or do they simply survive better in adverse conditions?


To address these questions, Goodell and Takahashi teamed up with Dr. Lawrence Donehower, professor of virology and microbiology at Baylor and co-author of the paper, who had been studying PPM1D for more than 20 years.

Survival in adversity
To understand what gives PPM1D mutants a competitive advantage, the researchers carried experiments in the lab mixing normal and PPM1D mutant cells together in a dish, growing them together and then exposing them to different environmental conditions. They were surprised to find that under normal conditions, PPM1D mutants and normal cells grew at the same rates. This suggests that the mutants are not intrinsically stronger. However, following exposure to cisplatin and other chemotherapy drugs, the researchers observed that PPM1D mutants dramatically outcompeted normal cells.

“Taking the results all together, our findings suggest that chemotherapy acts as an evolutionary selection pressure that favors the survival of PPM1D mutant cells because they have better fitness than normal cells and ‘win’ under this specific type of stress,” said Joanne Hsu, a student in the Medical Scientist Training Program (MD/PhD) at Baylor and a member of the Goodell lab. “So, when a patient receives cisplatin-based chemotherapy, stem cells carrying PPM1D mutations survive better. This growth advantage could provide fertile ground for subsequent acquisition of mutations that eventually lead to the development of secondary leukemia years later. This study has been a translational journey from bedside observations to bench side.”
“I am most excited that the findings from clinical samples at MD Anderson generated a hypothesis that was verified in Dr. Goodell’s lab,” Takahashi said. “This study is a great representation of how combined forces between clinical and basic science investigators from the two institutions can disclose an important scientific discovery.”
Video credit: Cell Press
The researchers’ findings in this study have multiple clinical implications for the future.
“Knowing that the selection of chemotherapeutic drugs can affect the risk of leukemia developing years later may assist physicians in the selection of specific treatments for their patients,” Goodell said.
Looking further ahead, this study also points towards PPM1D as a promising therapeutic target in patients with secondary leukemia.
“I am excited that this study shows that there is a need to develop therapeutic approaches to target overactive PPM1D signaling,” Donehower said.
Interested in reading all the details of this work? Visit the journal Cell Stem Cell.
Other contributors to this work include T. Dayaram, E. De Braekeleer, M. Jeong, F. Wang, J. Zhang, T. Hefferman, S. Gera, J. Kovacs, J. Marszalek, C. Bristow, Y. Yan, G. García-Manero, H. Kantarjian, G. Vassiliou and P.A. Futreal. The authors are associated with Baylor College of Medicine, the University of Texas MD Anderson Cancer Center, Wellcome Sanger Institute, University of Cambridge and the University of Texas Health Science Center at Houston.
Support for this study was provided by the Cancer Prevention and Research Institute of Texas (RP160451 and R120501) and the National Institutes of Health (DK092883, DK116428, S10RR024574, AI036211, P30 CA 125123 and P30 CA016672). Further support was provided by the Welch Foundation (G-0040), MD Anderson’s MoonShot Program, the Baylor Research Advocates for Student Scientists, the BCM MSTP program, the Khalifa Pyhisician Scientist Award, the Physician Scientist Program at MD Anderson and a Leukemia SPORE Cancer Enhancement Award. Additional support came from the Cancer Research UK Senior Cancer Research Fellowship (C22324/A23015), the Kay Kendall Leukaemia Foundation, Bloodwise, the Sanger Institute (WT098051) and the Samuel Waxman Cancer Research Foundation.