Mouse model helps solve 15q13.3 microdeletion puzzle
Individuals with a complex neurological condition called 15q13.3 microdeletion syndrome are missing a small piece of chromosome 15 that usually contains six genes. Although this information has significantly narrowed down the location of the genetic culprit, the challenge still remains for researchers to identify the gene within the deleted region that accounts for the clinical characteristics observed in patients.
“Which one of the genes is responsible for the clinical characteristics of patients has not been clear, and is very important,” said senior author Dr. Christian Schaaf, who was assistant professor of molecular and human genetics at Baylor College of Medicine during the development of this work, and currently is a visiting professor at Baylor and professor of clinical genomics at University Clinic Cologne, Germany. “If we want to get to the point where we can treat patients, we need to know which gene or genes to target. That is the big picture question behind this study.”

Patients with 15q13.3 microdeletion syndrome can present with a wide spectrum of neurodevelopmental disorders, including developmental delay, intellectual disability, epilepsy, language impairment, abnormal behaviors, neuropsychiatric disorders and hypotonia. Schaaf and his colleagues have studied this syndrome for several years.
“We have searched for the gene in question from different standpoints using different kinds of technology,” Schaaf said. “One way is with mouse models. We genetically engineer mice to lack a certain gene, and determine whether the animals show any features similar to what we see in the patients. For example, if the patients have epilepsy, we knockout a gene we suspect is involved in epilepsy and determine whether the mice also have the condition.”
Finding two new pieces of a complex genetic puzzle
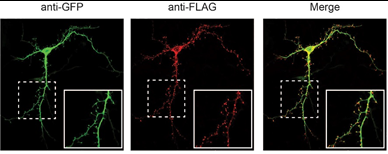
Of the six genes usually deleted in this syndrome, the gene OTUD7A was considered a strong candidate. This gene is one of only two genes that are always involved in the deletion, according to hundreds of patients studied by Schaaf and his colleagues. To test the relevance of the suspect gene, the researchers genetically engineered mice to lack this gene and observed that the mice indeed had many of the characteristics present in patients. In addition, they discovered that the gene is expressed in dendritic spines and that mice without the gene have fewer dendritic spines than those with it.
“We now have two new pieces of this complex genetic puzzle,” Schaaf said. “We found that the gene Otud7a seems to play a major role in many of the neurological problems observed in our mouse model, and that this gene could be considered a therapeutic target for the human condition.”
The researchers also learned that this gene is involved in the regulation of dendritic spine density. Dendritic spines are small protrusions along various types of neurons. They receive input from other neurons and participate in important neuronal fucntions.
Altogether, our findings suggest that a deficiency in the gene OTUD7A is a major contributor to the clinical characteristics associated with the 15q13.3 microdeletion syndrome via changes in the number of dendritic spines and their activity,” Schaaf said.
“We think that the Otud7a knockout mouse line we have engineered would be a valuable resource to other researchers who wish to study this condition further. We have deposited it with the Jackson Laboratory for immediate availability,” he said.
Find all the details about this study in the American Journal of Human Genetics.
Learn more about the research conducted in the Schaaf lab.
Other contributors to this work include Jiani Yin, Wu Chen, Eugene S. Chao, Sirena Soriano, Li Wang, Wei Wang, Steven E. Cummock, Huifang Tao, Kaifang Pang, Zhandong Liu, Fred A. Pereira, Rodney C. Samaco, Huda Y. Zoghbi and Mingshan Xue. The contributors are affiliated with Baylor College of Medicine and/or Texas Children’s Hospital.
The work was supported in part by the BCM Intellectual and Developmental Disabilities Research Center Neuroconnectivity, Neurovisualization and Neurobehavioral Cores (NIH Grant U54HD083092), the Baylor College of Medicine Genetically Engineered Mouse Core (NIH Grants P30CA125123, U42HG006352), NIH Grant DP5OD009134, NIH Grant R01NS100893, the Whitehall Foundation and Citizens United for Research in Epilepsy, a Caroline DeLuca Scholarship and the Joan and Stanford Alexander Family.