Dissemination of bone metastasis linked with bone remodeling
Years to decades after breast tumors have been removed, cancer may return or metastasize in other organs. Bone is frequently affected by metastasis and in the current study published in the journal Cancer Discovery, the lab of Dr. Xiang H.-F. Zhang at Baylor College of Medicine and colleagues show in an animal model that the initiation of bone metastasis is coupled with the normal bone repair process. The findings offer new directions for future studies to prevent tumor recurrence.

Awakening metastatic seeds
“As cancer progresses, a few cells may leave the primary tumor and travel to the bone, but we still don’t completely understand what determines the fate of these cells – we call them metastatic seeds,” said Zhang, William T. Butler, M.D., Endowed Chair for Distinguished Faculty and interim director of the Lester and Sue Smith Breast Center at Baylor.
Many metastatic seeds will die while others may remain inactive, and the patient will be asymptomatic after the primary tumor has been removed, Zhang explained. “But in a substantial number of patients the cancer comes back after some time,” said Zhang, a professor of molecular and cellular biology, a McNair scholar and a member of the Dan L Duncan Comprehensive Cancer Center at Baylor.
Research has begun to unveil the events that might trigger the awakening of dormant metastatic seeds. Previously, Zhang and his colleagues observed that metastatic seeds tend to concentrate where osteogenic cells are located. Osteogenic cells carry out the process of building new bone.
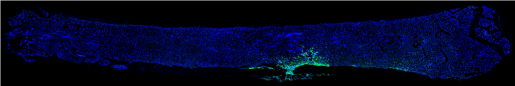
The bone is a very dynamic organ. It is remodeled every day. If there is a bone fracture, the process is even more dramatic. Injured bone is broken down and new bone grows to repair the injury.
“Seeing that metastatic seeds reside next to osteogenic cells made us wonder whether there could be a relationship between bone metastasis and bone repair,” Zhang said.
Working with a mouse model, the team investigated the effect of triggering the bone repair process on bone metastasis. They found that activating the process resulted in significantly more metastases.
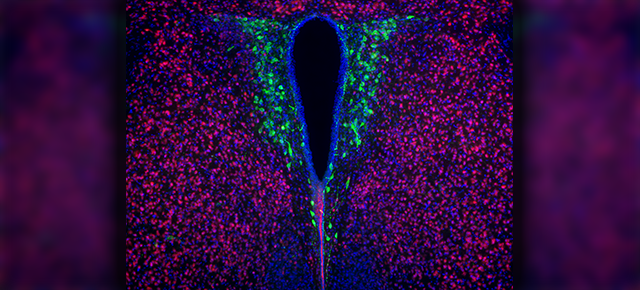
Furthermore, they discovered a subset of bone stem cells that express the marker NG2, called NG2-positive (NG2+) cells. These cells, which have long been known to drive normal bone turnover, seem to be directly involved in the awakening of metastatic seeds. The researchers showed that when they genetically removed the NG2 marker from these cells, there were fewer metastases.
“Before bone repair is triggered, say by an injury, both cell types, NG2+ and cancer cells, are dormant. But when the bone needs to be repaired, there is a signal that triggers bone turnover,” Zhang said.
NG2+ stem cells sense this signal and respond by migrating to the place where bone needs to be rebuilt and differentiating into cells that conduct the repair. “Interestingly, we discovered that cancer cells can physically adhere to the NG2+ cells via cell adhesion molecules,” Zhang said.
These findings suggest that cancer cells may ‘ride’ NG2+ cells, which would carry them to remodeling sites where, taking advantage of the differentiation and migration processes, they may transform from dormant to actively dividing cancer cells that may give rise to new tumors.
“This work opens new ways of thinking about how to prevent bone metastasis,” Zhang said.
The cancer cells are already in the bone but interfering with their association with NG2+ cells may one day help to stop or control bone metastasis.”
Other contributors to this work include: first author Weijie Zhang, Zhan Xu, Xiaoxin Hao, Tiancheng He, Jiasong Li, Yichao Shen, Kai Liu, Yang Gao, Jun Liu, David G. Edwards, Aaron M. Muscarella, Ling Wu, Liqun Yu, Longyong Xu, Xi Chen, Yi-Hsuan Wu, Igor L. Bado, Yunfeng Ding, Sergio Aguirre, Hai Wang, Zbigniew Gugala, Robert L. Satcher and Stephen T.C. Wong. The authors are affiliated with one or more of the following institutions: Baylor College of Medicine, Houston Methodist Cancer Center, University of Texas Medical Branch-Galveston and University of Texas MD Anderson Cancer Center-Houston.
This work was supported by the US Department of Defense DAMD W81XWH-16-1-0073 (Era of Hope Scholarship), NCI CA183878, NCI CA251950, DAMD W81XWH-20-1-0375, Breast Cancer Research Foundation, and McNair Medical Institute. Further support was provided in part by NCI U01CA252553, John S. Dunn Research Foundation, and T.T. and W.F. Chao Center for BRAIN. This project was supported by the RNA In Situ Hybridization Core with funding from the NIH (1S10OD016167), the Cytometry and Cell Sorting Core with funding from the CPRIT Core Facility Support Award (CPRIT-RP180672), the NIH (P30 CA125123, S10 RR024574, S10 OD025251), the Pathology Core of Lester and Sue Smith Breast Center, the Optical Imaging & Vital Microscopy Core and the Integrated Microscopy core at Baylor College of Medicine.



This is an interesting insight.
Kudos to the team!