How the egg and the embryo actually travel through the fallopian tube
Ask a scientist working on animal reproduction how the egg and the embryo make their way through the fallopian tube toward the uterus and the most likely response would be that the cells chug along slowly over the course of days.
In truth, before this work, nobody had actually seen the egg and the embryo travelling along the fallopian tube in live organisms, mainly because the location had been inaccessible for direct imaging.
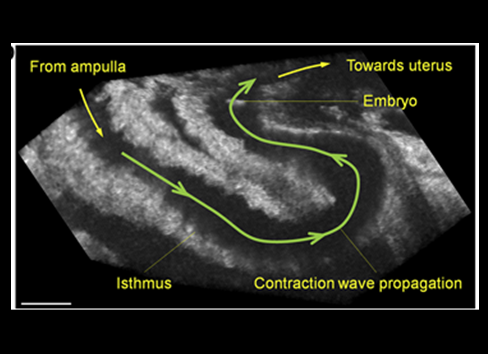
Looking to shed new light on the dynamics of the eggs prior to fertilization and embryo transport preceding implantation, Dr. Irina Larina at Baylor College of Medicine and Dr. Shang Wang at Stevens Institute of Technology developed a novel imaging approach that has allowed them to see eggs and embryos as they move along the fallopian tube in a live animal.
An accidental project
“I would call this an accidental project,” said Larina, associate professor of molecular physiology at Baylor and corresponding author of the work. “A colleague was writing a book on the development of the reproductive system in mice and asked us for a microscopy image. As we proceeded to capture an image, we saw an embryo inside the fallopian tube. The embryo was moving. We didn’t expect to see that, but triggered our curiosity. Could we find a way to film an egg and embryo as they travelled to the uterus?”

Larina and Wang first developed and combined intravital window and optical coherence tomography to have visual access to eggs and embryos as they are transported through the mouse oviduct.
With their new imaging techniques they discovered several unexpected findings. The expectation was that murine eggs and embryos after fertilization would move very slowly through roughly a 1 inch-long fallopian tube over the course of about three days. The accepted idea was that hair-like structures called cilia, which line the internal surface of the fallopian tube, mediated the movement of the cells.
An unexpected journey supports reevaluating previous knowledge
“Surprisingly, we found that eggs and embryos move along the fallopian tube by combining different types of movements, showing a dynamic process that is more complex than it had been thought until now,” said first author Wang, assistant professor in the Department of Biomedical Engineering at Stevens Institute of Technology.

Depending on the location along the fallopian tube, the cells were observed sometimes moving in fast circular movements or oscillating, moving back-and-forth over long distances or fast forward. The orchestration of these different movements involves the participation of cilia, muscle contractions and peristaltic movements, processes that are differentially regulated by hormones and other factors.
“Our findings provide a better understanding of this important step in mammalian reproduction and support reevaluating previous knowledge about how it happens,” Larina said. “In addition, our observations imply that perturbing one or more of the different movements along the tube could lead to reproductive disorders.”
“Applying our imaging approach can result in exciting discoveries that we hope can advance our understanding of disordered human reproductive conditions associated with the fallopian tube, as well as improvements in in vitro fertilization,” Wang said.
Read all the details and see the images and videos in the paper published in Cell Reports.
This video shows separation of an individual embryo from a large embryo group in the lower ampulla of the mouse oviduct in vivo. Courtesy of the authors/Cell Reports, 2021.
This study was supported by NIH with grants (R21EB028409, R01HL120140, R01EB027099 and R01HD096335) as well as the Start-Up Funds from Stevens Institute of Technology.
Visit the Larina lab website to learn how they apply optical engineering and computational modeling and analysis to understand mechanisms regulating normal and abnormal developmental processes in animal models.