Researchers discover novel regulators of myelin repair and regeneration
Rapid and accurate transmission of nerve signals highly depends on the integrity of myelin, an insulating sheath that surrounds the nerves of the brain and the spinal cord.
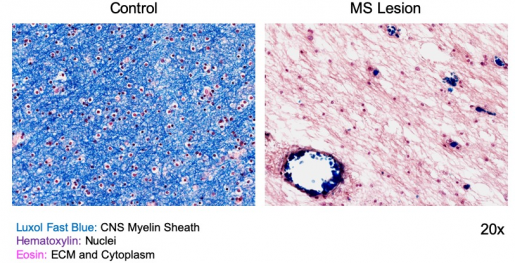
Damage or loss of the myelin sheath is a hallmark of several neurological diseases in adults (such as multiple sclerosis, MS, in the image below), as well as in infants (cerebral palsy, for instance), and is common after injury. However, little is known about how cells repair and regenerate myelin.
Dr. Hyun Kyoung Lee, assistant professor of pediatrics at Baylor College of Medicine and the Jan and Dan Duncan Neurological Research Institute at Texas Children’s Hospital, and her colleagues identified a new regulator (Nedd4) and a genetic pathway (Daam2- Nedd4-VHL) that control myelin formation during development and after injury. Their findings offer the exciting possibility of therapeutically targeting this new pathway to repair and regenerate damaged myelin after an injury or in patients suffering from myelin-associated neurological disorders.

Taking a closer look at myelin production
“Myelin is produced and assembled into sheaths by glial cells called oligodendrocytes (OL) via a series of steps regulated by signaling pathways,” said Lee, who also is a member of Baylor’s Dan L Duncan Comprehensive Cancer Center. “While several key features are common between developmental and regenerative myelination, it remains unclear how developmental programs are re-activated after an injury to initiate repair and regeneration in adulthood.”
Several years back, Lee and others in Dr. Benjamin Deneen’s lab discovered a regulator, called dishevelled associated activator of morphogenesis 2 (Daam2), that suppressed OL development and repair. Interestingly, the researchers’ recent work in glial cancers led them to the discovery that Daam2 interacted with the hypoxia-associated von Hippel-Lindau (VHL) ubiquitin ligase.
“We found that in glial tumors (glioma), Daam2 and VHL displayed an antagonistic relationship – Daam2 promoted the degradation of VHL,” Lee said. “Intrigued by this finding, we wondered whether Daam2-VHL displayed similar interactions in the context of myelin development and repair and this became our impetus for the current study.”
They found that Daam2 indeed suppressed VHL expression and reduced its stability during OL development and regeneration, and showed that VHL acts downstream of Daam2 during glial development.
Further studies showed that Daam2-mediated degradation of VHL involved the ubiquitin-proteosomal system (UPS), which is a crucial protein-degradation system found in the cells of most multicellular organisms. E3 ubiquitin ligases are a group of important enzymes of the UPS that specifically recognize the target protein and catalyze the transfer of ubiquitin molecules that mark the target for further steps of degradation.
To understand how Daam2 promotes the degradation of VHL, the team performed a proteomic screen to identify the candidate ubiquitin ligase(s). After several rounds of screening and validation, they narrowed down Nedd4 as the target ubiquitin ligase.
This was an exciting discovery because, although Nedd4 has been implicated in early neuronal development and cancers, this was the first demonstration of its role in glial development and regeneration,” Lee said.
“We showed that Nedd4 is necessary and sufficient for developmental and regenerative myelination.”
Consistent with this newly identified pro-myelination role for VHL and Nedd4, they saw a dramatic reduction in the levels of VHL or Nedd4 in the post-mortem brain tissues obtained from patients with multiple sclerosis.
“SiNce VHL and Nedd4 are already known to play important roles in several cancers, we think down the road our findings could have broader implications that may well extend beyond myelin-associated neurological disorders or white matter injury to include malignancies,” Lee concluded.
Read all the details of this work in the journal Genes & Development.
Other authors include Xiaoyun Ding, Juyeon Jo, Chih-Yen Wang, Carlo D. Cristobal, Zhongyuan Zuo, Qi Ye, Marvin Wirianto, Aaron Lindeke-Myers, Jong Min Choi, Carrie A. Mohila, Hiroshi Kawabe, Sung Yun Jung, Hugo J. Bellen, and Seung-Hee Yoo. They are affiliated to one or more of the following institutions: Baylor College of Medicine, Texas Children’s Hospital, University of Texas Health Science Center and Max Planck Institute of Experimental Medicine.
This work was supported by grants from the National Multiple Sclerosis Society (RG-1907-34551), the Hilton Foundation (#17328), National Institutes of Health (NIH)/NINDS (R01NS110859-01), the Welch Foundation (AU-1971-20180324) and NIH/NIGMS (R01GM114424).