Brain tumors trigger silencing of neuronal activity
Sudden episodes of confusion and seizures often are the first signs of a malignant brain tumor. At Baylor College of Medicine, Dr. Jeffrey Noebels, director of the Blue Bird Circle Developmental Neurogenetic Laboratory, and his colleagues want to understand how tumor cells alter the excitability of brain circuits as this is crucial to finding comprehensive cures.
“Since seizures may persist even after surgical removal of the tumor and tumors may become resistant to medication or even promote the regrowth of malignant cells, being able to identify why and how these early neurological changes occur is needed,” said Noebels.

The findings, which appear in the Journal of Clinical Investigation, are a collaboration between scientists in the Blue Bird Circle Developmental Neurogenetic Laboratory in the Department of Neurology and the Center for Cell and Gene Therapy at Baylor.
How tumors can silence neuronal activity
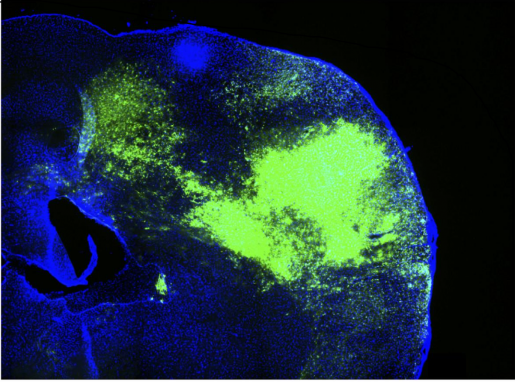
Using glioma mouse models, researchers detected waves of ultra-slow depolarization arising from the tumor margin. Depolarization occurs when cells change their electric charge distribution, a process that is essential to communication between cells, among other functions. The tumor margin is the border where tumor tissue meets healthy cells. These waves of ultra-slow depolarization were found to spread across the cortical surface to transiently silence all neuronal activity for several minutes.
Depolarizing waves that slowly advance across the brain, silencing neuronal firing and then fading away, were first described by the physiologist Aristedes Leão in the 1940s. It has been linked to brief spells of blindness during the visual aura in migraine headaches and linked to paralysis on one side of the body when the wave silences the motor system. They are triggered by high levels of local glutamate and potassium signaling or low levels of oxygen. When prolonged, it may injure underlying brain cells.
In the current study, researchers detected the depolarizing waves earlier than previously found, during a reproducible progression of inhibitory cell loss, glutamate dysregulation and inflammation in the tumor margin, and found that this excitability progressed more slowly when the tumor grew in a host brain that is genetically resistant to seizures.
Unlike previous experimental models that inject human tumor cells into an immunocompromised adult mouse brain, the Baylor tumor group involved in the current study, led by Dr. Benjamin Deneen, professor of neurosurgery, and Dr. Kwanha Yu, assistant professor, both in the Center for Stem Cell and Regenerative Medicine, genetically engineered the model by injecting human tumor gene mutations into a mouse embryo with a normal immune system.
These mice develop a reproducible glioma resembling the human tumor progression. This model allowed the Noebels team to study the serial evolution of tumor hyperexcitability over several months in this more naturalistic model. Noebels’ team was made up of Asante Hatcher, Baylor graduate student, Dr. Isamu Aiba, assistant professor of neurology and Dr. Jochen Meyer, assistant professor of neurology, all at Baylor.
While large tumors can raise intracranial pressure and cause headache at advanced stages, pressure alone does not account for the earliest problems commonly encountered by patients, such as ‘tumor spells’ of confusion, speech arrest and motor seizures,” said Noebels.
“Identifying this early tumor biomarker in a more naturalistic model may be helpful in assessing the status of neocortex following tumor removal and guide antiepileptic therapy development for brain tumor-related epilepsy,” said Noebels, who also is professor neurology, neuroscience, and molecular and human genetics at Baylor.
The study was funded by the NIH National Cancer Institute.