Some antibiotic-resistant bacteria have outside help
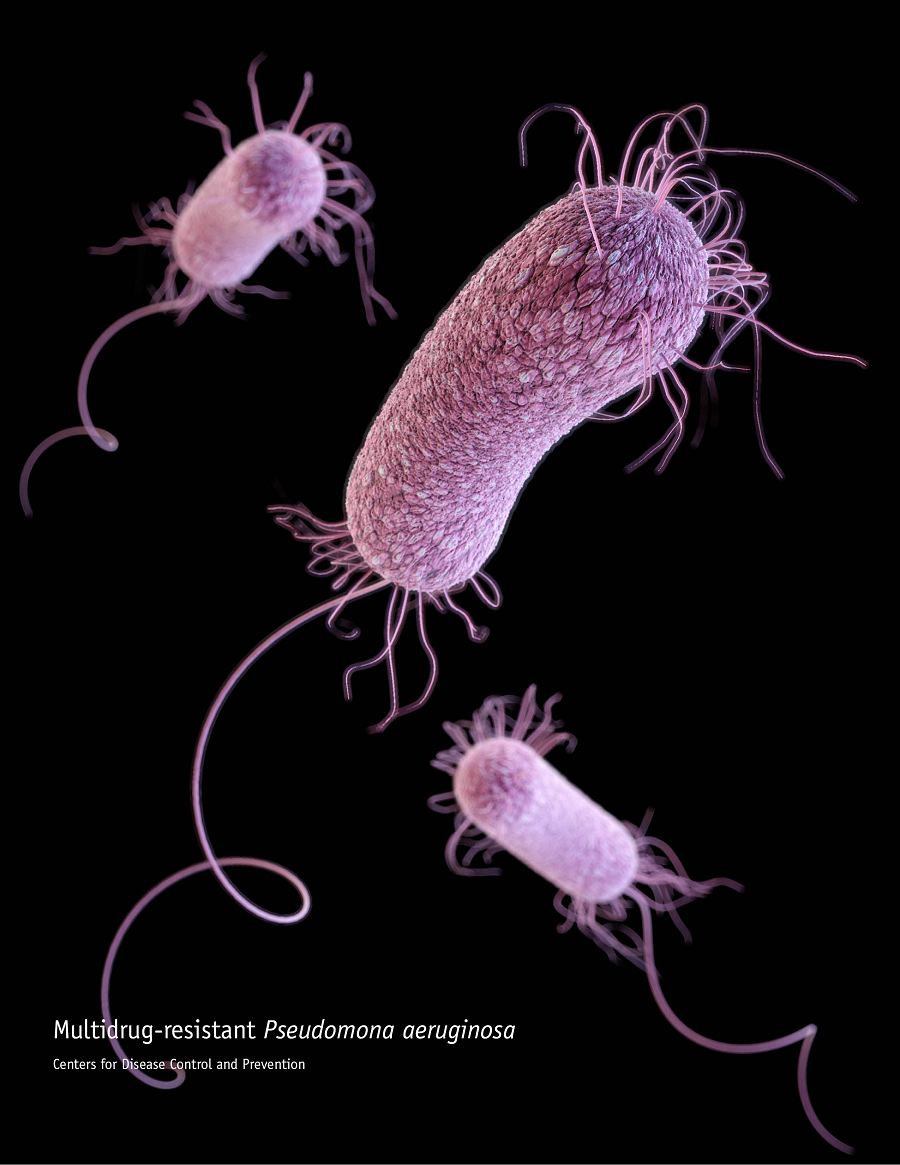
The bacterium Pseudomonas aeruginosa – one of the greatest threats to human health, according to the World Health Organization – frequently carries a virus or phage called Pf. The two have forged an unsuspected alliance that gives bacteria the upper hand when establishing an infection.
The problem
At Baylor College of Medicine, Dr. Sundeep Keswani, professor of surgery and surgical director of basic science research at Texas Children’s Hospital, and Dr. Swathi Balaji, assistant professor of surgery, study the regulation of physiological wound healing.
In this work, they joined their long-time collaborator, Dr. Paul L. Bollyky, assistant professor of medicine- infectious diseases and of microbiology and immunology at Stanford University, and other researchers to further investigate Bollyky’s finding that P. aeruginosa’s Pf phage increases the bacteria’s ability to infect people.
“In chronic non-healing wounds, P. aeruginosa is the ring leader preventing wounds to heal,” Keswani said. “These increasingly drug-resistant bacteria also infect the lungs of most adults with cystic fibrosis and cause a substantial portion of all infections of diabetic ulcers, bedsores and burn wounds. Infected diabetic foot ulcers are the single largest cause of amputation.”

The arsenal clinicians use to fight P. aeruginosa is limited. A progressively shorter list of effective antibiotics and no approved vaccines highlight the urgency of finding new ways to prevent or control this type of infections.
In chronic wound infections, P. aeruginosa forms biofilm-like clusters. Previous work found that Pf phage interacts with the biofilm inducing a liquid crystal matrix that boosts the bacteria’s tolerance to desiccation and antibiotics. This and other evidence from animal models of P. aeruginosa infection suggested that Pf phage may contribute to the establishment of chronic infections and may help P. aeruginosa evade host defense mechanisms.
Team work
In this study, the researchers looked closely into how the bacterium/phage team managed to prevent clearance of bacterial infections. They found Pf phages frequently in human bacterial infections; they are present in 68 percent of wounds that are positive for P. aeruginosa. Then, to investigate whether Pf phage could actually help P. aeruginosa establish an infection, they worked with Keswani and Balaji to establish a mouse model of wound infection and develop appropriate methods to evaluate healing in these infected wounds.
“In our mouse model of wound infection we use a luminescent P. aeruginosa and quantify the level of bacterial infection by measuring luminescence in the wounds,” Keswani said. “We compared the level of infection caused by bacteria with and without the phage and found that having the phage gave the bacteria a great advantage.”
To cause an infection, the dose of P. aeruginosa without the phage had to be 50 times greater than the dose of bacteria with the phage.”
Using this model, the researchers also found that more mice died when they were infected with the bacteria/phage combination than with bacteria alone. Together, these experiments showed that Pf phage promotes both incidence and virulence of P. aeruginosa in wound infections.
Changing the plan of attack
To add further support to the evidence that the Pf phage promotes P. aeruginosa infection, the researchers tested the effect of antibodies against the phage on the mouse model of infection.
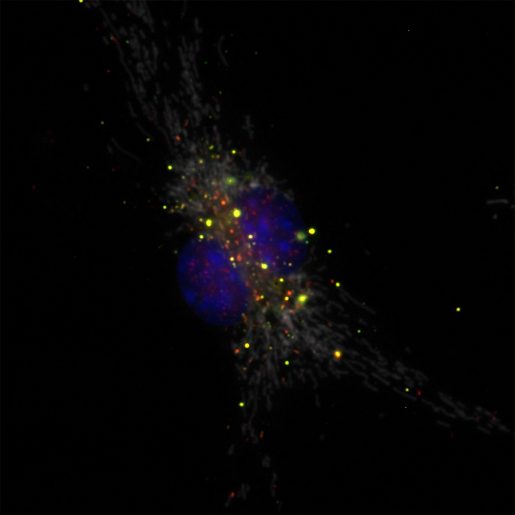

“Adding antibodies directed against the phage in the wounds with bacteria reduced wound infection significantly,” Balaji said. “Importantly, these results indicate that antibodies against the phage can protect from P. aeruginosa infection, suggesting new strategies to fight back this serious public health problem, such as developing a vaccine against the phage.”
The researchers also investigated the mechanism by which Pf phage helps bacteria establish an infection. Using cell cultures in the lab they discovered that Pf phages present in wounds are eaten by macrophages, a type of immune cell that also participates in developing an immune response to clear P. aeruginosa infection. Ingesting the phage triggered a change in the macrophages’ activity; they switched from fighting the bacteria to fighting the virus. Uncontested, the bacteria proliferated maintaining an infection that did not heal.
“It’s an exciting time for the wound healing field,” Keswani said.
We have been treating bacterial infections by trying to kill the bacteria. Now we know that we may have the upper hand if we target the phages that are inhibiting the immune response toward bacteria, and also fight the bacteria with antibiotics.”
“In addition,” Balaji said, “we can address these paradigm shifting questions in our labs thanks to the cutting-edge imaging and other Advanced Technology Core facilities that we have here at Baylor.”
Interested in reading all the details of this work? Find it in the journal Science.
Other institutions involved in this project include Palo Alto Veterans Institute of Research, the University of Washington and the University of Montana.
For a complete list of contributors, their affiliations and financial support for this study, follow this link.