Following up with long-term survivor of CAR T cell therapy
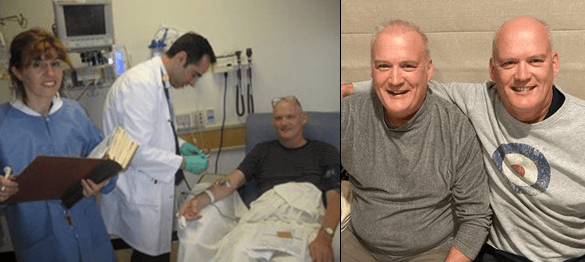
Matthew Rea was diagnosed with non-Hodgkin’s lymphoma in July 2009. He had been feeling sick for a while, but he assumed it was due to stress at work. By the time he received his diagnosis, the cancer had already progressed to stage four. The New Zealand native started an aggressive treatment plan of chemotherapy and immunotherapy right away. If everything went as planned, he could follow that treatment with a stem cell transplant, which would provide the best chance for remission. He already had a donor in the family – his identical twin brother.
Meanwhile, he continued to explore other treatment options. He contacted friends at Baylor College of Medicine to discuss his treatment, and they recommended a clinical trial for a new type of immunotherapy called CAR T cell therapy. The treatment involves harvesting the patient’s own T cells – or in Rea’s case, his twin brother’s – and modifying them to produce chimeric antigen receptors, which recognize and attack certain proteins on cancer cells. Researchers at the Center for Cell and Gene Therapy at Baylor, Texas Children’s Hospital and Houston Methodist Hospital developed the therapy used in the trial. It targets the CD19 protein, which is highly expressed in B cell lymphomas like non-Hodgkin’s lymphoma.
Rea decided it was worth an international trip to give the treatment a chance. In 2010, six weeks after he received a stem cell transplant, he and his wife flew to Houston for the CAR T infusion. He was the first patient to receive syngeneic (i.e. made from an identical twin) CD19 CAR T cells. Dr. Carlos Ramos, a specialist in cellular therapy at Houston Methodist Hospital, infused the treatment. More than a decade later, he remembers the significance of that clinical trial.
“There were very few reported cases of patients treated with these types of immune cell products at the time, and we had a mix of excitement and anxiety over administering them,” said Ramos, professor in the Center for Cell and Gene Therapy and member of the Dan L Duncan Comprehensive Cancer Center at Baylor. “We were hoping to have a beneficial effect on the patients that we were treating, but we also realized that these were first-in-man studies and we wanted to be ready to detect any problem immediately. Matt was a great sport and put up with our frequent queries.”
Rea remembers the treatment as easy compared to his rounds of chemotherapy. He felt no side effects as a result of the CAR T cell therapy. To this day, Rea remains cancer free. He has experienced no relapses or other side effects following his treatment.
“I felt like I was given a lifeline to come to Houston, and I feel incredibly privileged to have been a part of that clinical trial,” Rea said. “This treatment gave me hope for a cure, not just temporary remission.”
Since Rea’s treatment, the FDA has approved autologous CD19-specific CAR T cells for the treatment of relapsed B-cell lymphomas, and the therapy is commercially available. According to Ramos, it has changed the paradigm for how these cancers are treated.
“A significant proportion of patients who had diseases that could not be controlled with conventional chemotherapy may now be able to be cured by this approach,” Ramos said. “Moreover, this treatment may be better than high-dose chemotherapy and autologous stem cell transplant, which is one of the strategies used to manage patients with relapsed lymphoma and that can have significant toxicities.”
CAR T cell therapy use has expanded widely in the last decade. Autologous CAR T cells targeting other antigens are starting to be used for other blood cancers, such as multiple myeloma, with encouraging results.
As the first person to receive syngeneic CD19 CAR T therapy and become a long-term survivor, Rea’s case is important for follow-up study. Baylor researchers have stayed in touch with him since his treatment to ensure the treatment did not cause any harmful side effects.
“Because the treatment involves the use of genetically modified immune cells, one of the main concerns is whether that genetic modification may lead to a secondary cancer,” Ramos said. “We have not seen that in any of our protocols, but there have been some recent reports suggesting that this is a possible, albeit rare, occurrence. If it does happen, we want to be sure that we get that information quickly, so that we can mitigate any problems promptly. These issues can occur several years after treatment and, therefore, we want to make sure that we follow these patients closely for a long time.”
For this reason, Rea has been sending vials of his blood regularly to Houston from New Zealand. Vicky Torrano, the study’s research nurse at the Center for Cell and Gene Therapy, is on the receiving end of these deliveries in Houston.
“The limited number of flights during the COVID pandemic made it extremely challenging to receive blood samples during those times,” Torrano said. “But we are back on track.”
“Every year, I look forward to receiving a FedEx package from Vicky with my collection kit,” Rea said. “I can’t believe I have been sending my blood back to Houston for more than a decade.”
Read more about long-term follow-up for recipients of genetically modified cells in this publication by Dr. Helen Heslop, director of the Center for Cell and Gene Therapy.
By Molly Chiu



